DOI:
https://doi.org/10.14483/23448393.23475Publicado:
2025-12-09Número:
Vol. 30 Núm. 3 (2025): Septiembre-diciembreSección:
Ingeniería CivilA Novel Rotational Limestone Treatment System for Effective Acid Mine Drainage Remediation
Un sistema novedoso de tratamiento rotacional de roca caliza para la remediación eficaz del drenaje ácido de minas
Palabras clave:
Coal mining, Acid mine drainage, rotational system, superficial degradation (en).Palabras clave:
Minería del carbón, drenaje ácido de mina, sistema rotacional, degradación superficial (es).Descargas
Referencias
[1] G. Naidu, S. Ryu, R. Thiruvenkatachari, Y. Choi, S. Jeong, and S. Vigneswaran, “A critical review on remediation, reuse, and resource recovery from acid mine drainage,” Environ. Poll., vol. 247, pp. 1110–1124, Apr. 2019. https://doi.org/10.1016/j.envpol.2019.01.085
[2] H. E. Ben Ali, C. M. Neculita, J. W. Molson, A. Maqsoud, and G. J. Zagury, “Performance of passive systems for mine drainage treatment at low temperature and high salinity: A review,” Miner. Eng., vol. 134, pp. 325–344, Apr. 2019. https://doi.org/10.1016/j.mineng.2019.02.010
[3] D. Silva, C. Weber, and C. Oliveira, “Neutralization and uptake of pollutant cations from acid mine drainage (amd) using limestones and zeolites in a pilot-scale passive treatment system,” Miner. Eng., vol. 170, art. 107000, Aug. 2021. https://doi.org/10.1016/j.mineng.2021.107000
[4] J. S. Pozo-Antonio, I. Puente-Luna, S. L. López, and M. V. Ríos, “Tratamiento microbiano de aguas ácidas resultantes de la actividad minera: una revisión,” Tecno. Cien. Agua, vol. 8, no. 3, pp. 75–91, 2017. https://doi.org/10.24850/j-tyca-2017-03-05
[5] I. Park et al., “A review of recent strategies for acid mine drainage prevention and mine tailings recycling,” Chemosphere, vol. 219, pp. 588–606, Mar. 2019. https://doi.org/10.1016/j.chemosphere.2018.11.053
[6] I. Labastida, M. A. Armienta, R. H. Lara, R. Briones, I. González, and F. Romero, “Kinetic approach for the appropriate selection of indigenous limestones for acid mine drainage treatment with passive systems,” Sci. Total Environ., vol. 677, pp. 404–417, Aug. 2019. https://doi.org/10.1016/j.scitotenv.2019.04.373
[7] D. M. Acosta-Bueno, “Impactos ambientales de la minería de carbón y su relación con los problemas de salud de la población del municipio de Samacá (Boyacá), según reportes ASIS 2005-2011,” Specialization dissertation, Dept. Ed. Sci., Udistrital, Bogotá, Colombia, 2016.
[8] C. UPME, Ministerio de Minas y Energías, “Plan nacional de desarrollo minero con horizonte a 2025: Minería responsable con el territorio,” 2017. [Online]. Available: https://www1.upme.gov.co/simco/PlaneacionSector/Documents/PNDM_Dic2017.pdf
[9] R. H. Garzón, “Minería del carbón en Boyacá: entre la informalidad minera, la crisis de un sector y su potencial para el desarrollo,” Zero, vol. 33, no. 2344–8431, 2015. [Online]. Available: https://zero.uexternado.edu.co/mineria-del-carbon-en-boyaca-entre-la-informalidad-minera-la-crisis-de-un-sector-y-su-potencial-para-el-desarrollo/
[10] C. A. Agudelo Calderón, J. C. García-Ubaqie, R. Robledo Martínez, C. A. García-Ubaque, and L. Quiroz-Arcentales, “Evaluación de condiciones ambientales: aire, agua y suelos en áreas de actividad minera en Boyacá, Colombia,” Rev. Salud Púb., vol. 18, no. 1, pp. 50–60, Apr. 2016. https://doi.org/10.15446/rsap.v18n1.55384
[11] A. L. Boyles et al., “Systematic review of community health impacts of mountaintop removal mining,” Environ. Int., vol. 107, pp. 163–172, Oct. 2017. https://doi.org/10.1016/j.envint.2017.07.002
[12] I. Moodley, C. M. Sheridan, U. Kappelmeyer, and A. Akcil, “Environmentally sustainable acid mine drainage remediation: Research developments with a focus on waste/by-products,” Miner. Eng., vol. 126, pp. 207–220, Sep. 2018. https://doi.org/10.1016/j.mineng.2017.08.008
[13] L. E. Bertassello, P. S. C. Rao, J. Park, J. W. Jawitz, and G. Botter, “Stochastic modeling of wetland-groundwater systems,” Adv. Water Resour., vol. 112, pp. 214–223, Feb. 2018. https://doi.org/10.1016/j.advwatres.2017.12.007
[14] C. Zipper, J. Skousen, and C. Jage, “Passive treatment of acid-mine drainage,” Virgina Cooperative Extension, 2018. [Online]. Available: https://www.pubs.ext.vt.edu/content/dam/pubs_ext_vt_edu/460/460-133/CSES-216.pdf
[15] H. L. Yadav and A. Jamal, “Removal of heavy metals from acid mine drainage: A review,” Int. J. New Technol. Sci. Eng., vol. 2, no. 3, pp. 77–84, 2015. ISSN 2349-0780. Available: https://www.ijntse.com/upload/1443503976hlyand%20ajamal%20sir%202015.pdf
[16] D. Bejan and N. J. Bunce, “Acid mine drainage: electrochemical approaches to prevention and remediation of acidity and toxic metals,” J. Appl. Electrochem., vol. 45, no. 12, pp. 1239–1254, Dec. 2015. https://doi.org/10.1007/s10800-015-0884-2
[17] J. G. Skousen, P. F. Ziemkiewicz, and L. M. McDonald, “Acid mine drainage formation, control and treatment: Approaches and strategies,” Extr. Ind. Soc., vol. 6, no. 1, pp. 241–249, Jan. 2019. https://doi.org/10.1016/j.exis.2018.09.008
[18] G. R. Watzlaf, K. T. Schroeder, and C. L. Kairies, “Long-term performance of anoxic limestone drains,” Mine Water Environ., vol. 19, no. 2, pp. 98–110, Sep. 2000. https://doi.org/10.1007/BF02687258
[19] J. Skousen et al., “Review of passive systems for acid mine drainage treatment,” Mine Water Environ., vol. 36, no. 1, pp. 133–153, Mar. 2017. https://doi.org/10.1007/s10230-016-0417-1
[20] J. E. Santos Jallath, F. M. Romero, R. Iturbe Argüelles, A. Cervantes Macedo, and J. Goslinga Arenas, “Acid drainage neutralization and trace metals removal by a two-step system with carbonated rocks, Estado de Mexico, Mexico,” Environ. Earth Sci., vol. 77, no. 3, p. 86, Feb. 2018. https://doi.org/10.1007/s12665-018-7248-2
[21] C. R. Blanco-Zuñiga, N. Rojas-Arias, L. Y. Peña-Pardo, M. E. Mendoza-Oliveros, and S. A. Martinez-Ovalle, “Study of the influence of clays on the transfer of dissolved oxygen in water,” Ingeniería, vol. 26, no. 1, pp. 1–8, 2021. https://doi.org/10.14483/23448393.15846
[22] Y. Bao, C. Guo, G. Lu, X. Yi, H. Wang, and Z. Dang, “Role of microbial activity in Fe(III) hydroxysulfate mineral transformations in an acid mine drainage-impacted site from the Dabaoshan Mine,” Sci. Total Environ., vol. 616–617, pp. 647–657, Mar. 2018. https://doi.org/10.1016/j.scitotenv.2017.10.273
[23] E. W. Rice, R. B. Baird, and A. Eaton, Standard methods for the examination of water and wastewater, 23rd ed. Washington DC, USA: American Public Health Association, American Water Works Association, Water Environment Federation, 2017. https://yabesh.ir/wp-content/uploads/2018/02/Standard-Methods-23rd-Perv.pdf
[24] J. Skousen, A. Sexstone, K. Garbutt, and J. Sencindiver, “Wetlands for treating acid mine drainage,” Green Lands, vol. 22, no. 4, pp. 31–39, 1992.
[25] P. F. Ziemkiewicz, J. G. Skousen, D. L. Brant, P. L. Sterner, and R. J. Lovett, “Acid mine drainage treatment with armored limestone in open limestone channels,” J. Environ. Qual., vol. 26, no. 4, pp. 1017–1024, Jul. 1997. https://doi.org/10.2134/jeq1997.00472425002600040013x
[26] L. Merrill and W. A. Bassett, “The crystal structure of CaCO3 (II), a high-pressure metastable phase of calcium carbonate,” Acta Crystallogr. B, vol. 31, no. 2, pp. 343–349, Feb. 1975. https://doi.org/10.1107/S0567740875002774
[27] P. L. Althoff, “Structural refinements of dolomite and a magnesian calcite and implications for dolomite formation in the marine environment,” Amer. Miner. J. Earth Planet. Mat., vol. 62, pp. 772–783, 1977.
[28] K. Dong, H. Morikawa, S. Iwai, and H. Aoki, “The crystal structure of magnesite,” Amer. Miner. J. Earth Planet. Mat., vol. 58, no. 11, pp. 1029–1033, 1973.
[29] H. d’Amour, W. Denner, and H. Schulz, “Structure determination of α-quartz up to 68 x 10^8 Pa,” Acta Crystallogr. B, vol. 35, no. 3, pp. 550–555, Mar. 1979. https://doi.org/10.1107/S056774087900412X
[30] J. A. N. Carlos, B. Barrios, M. Lucia, M. Castro, and S. M. Arenas, “Prospectiva estratégica en los procesos de extracción y del beneficio de la roca caliza en el norte del Cesar, Colombia,” Rev. Agunkuya, vol. 4, pp. 1–14, 2018. https://digitk.areandina.edu.co/server/api/core/bitstreams/d98c24ee-bd50-45b6-a541-cd80904882ce/content
[31] G. R. Watzlaf, K. T. Schroeder, and C. L. Kairies, “Long-term performance of anoxic limestone drains,” Mine Water Environ., vol. 19, no. 2, pp. 98–110, Sep. 2000. https://doi.org/10.1007/BF02687258
[32] Ministerio de Ambiente y Desarrollo Sostenible, “Resolución 0631 De 2015. Diario Oficial No. 49.486 de 18 de abril de 2015,” 2015. [Online]. Available: https://www.minambiente.gov.co/wp-content/uploads/2021/11/resolucion-631-de-2015.pdf
[33] L. Bernier, M. Aubertin, A. M. Dagenais, B. Bussière, L. Bienvenu, and J. Cyr, “Limestone drain design criteria in AMD passive treatment: theory, practice and hydrogeochemistry monitoring at Lorraine Mine Site, Temiscamingue,” CIM Minespace 2001 Québec, 2001. [Online]. Available: https://www.researchgate.net/profile/Louis-Bernier-3/publication/265411811_Limestone_Drain_Design_Criteria_in_AMD_Passive_Treatment_Theory_Practice_and_Hydrogeochemistry_Monitoring_at_Lorraine_Mine_Site_Temiscamingue/links/5757fc9e08aef6cbe3626507/Limestone-Drain-Design-Criteria-in-AMD-Passive-Treatment-Theory-Practice-and-Hydrogeochemistry-Monitoring-at-Lorraine-Mine-Site-Temiscamingue.pdf
[34] L. N. Plummer, D. L. Parkhurst, and T. M. L. Wigley, “Critical review of the kinetics of calcite dissolution and precipitation,” in Chemical Modeling in Aqueous Systems, New York, NY, USA: ACM, 1979, ACM Symposium Series, vol. 93, pp. 537–573. https://doi.org/10.1021/bk-1979-0093.ch025
[35] W. A. M. Fernando, I. M. S. K. Ilankoon, T. H. Syed, and M. Yellishetty, “Challenges and opportunities in the removal of sulphate ions in contaminated mine water: A review,” Miner. Eng., vol. 117, pp. 74–90, Mar. 2018. https://doi.org/10.1016/j.mineng.2017.12.004
[36] O. Aduvire, Drenaje acido de mina: generación y tratamiento. Madrid, Spain: Instituto Geológico y Minero de España Dirección de Recursos Minerales y Geoambiente, 2006.
[37] T. V. Rakotonimaro, C. M. Neculita, B. Bussière, T. Genty, and G. J. Zagury, “Performance assessment of laboratory and field-scale multi-step passive treatment of iron-rich acid mine drainage for design improvement,” Environ. Sci. Poll. Res., vol. 25, no. 18, pp. 17575–17589, Jun. 2018. https://doi.org/10.1007/s11356-018-1820-x
[38] M. Jouini, T. V. Rakotonimaro, C. M. Neculita, T. Genty, and M. Benzaazoua, “Stability of metal-rich residues from laboratory multi-step treatment system for ferriferous acid mine drainage,” Environ. Sci. Poll. Res., vol. 26, no. 35, pp. 35588–35601, Dec. 2019. https://doi.org/10.1007/s11356-019-04608-1
[39] B. A. Wills, “Grinding Mills,” in Mineral Processing Technology, 4th ed. Amsterdam, Netherlands, Elsevier, International Series on Materials Science and Technology, 1988, ch. 7, pp. 253–308. https://doi.org/10.1016/B978-0-08-034937-4.50016-8
[40] D. K. Nordstrom, D. W. Blowes, and C. J. Ptacek, “Hydrogeochemistry and microbiology of mine drainage: An update,” App. Geochem., vol. 57, pp. 3–16, Jun. 2015. https://doi.org/10.1016/j.apgeochem.2015.02.008
[41] R. L. Pendleton and D. Nickerson, “Soil colors and special Munsell soil color charts,” Soil Sci., vol. 71, no. 1, pp. 35–44, 1951.
[42] E. Iakovleva, E. Mäkilä, J. Salonen, M. Sitarz, S. Wang, and M. Sillanpää, “Acid mine drainage (AMD) treatment: Neutralization and toxic elements removal with unmodified and modified limestone,” Ecol. Eng., vol. 81, pp. 30–40, Aug. 2015. https://doi.org/10.1016/j.ecoleng.2015.04.046
[43] Y. Chai et al., “Experimental study and application of dolomite aeration oxidation filter bed for the treatment of acid mine drainage,” Miner. Eng., vol. 157, art. 106560, Oct. 2020. https://doi.org/10.1016/j.mineng.2020.106560
[44] J. Demchak, T. Morrow, and J. Skousen, “Treatment of acid mine drainage by four vertical flow wetlands in Pennsylvania,” Geochem. Explor. Environ. Analysis, vol. 1, no. 1, pp. 71–80, Feb. 2001. https://doi.org/10.1144/geochem.1.1.71
[45] F. Scholz and H. Kahlert, “The calculation of the solubility of metal hydroxides, oxide-hydroxides, and oxides, and their visualisation in logarithmic diagrams,” ChemTexts, vol. 1, no. 1, p. 7, Mar. 2015. https://doi.org/10.1007/s40828-015-0006-0
[46] H. A. Peláez Morales, M. C. Prada Fonseca, G. Caicedo Pineda, C. X. Moreno Herrera, and M. A. Márquez Godoy, “Influence of the initial relation of Fe3+/Fe2+ in the process of biodesulfuration of a coal sample solution,” Rev. Int. Cont. Amb., vol. 29, no. 2, pp. 2111–217, 2013. ISSN 0188-4999. Available: https://www.scielo.org.mx/pdf/rica/v29n2/v29n2a7.pdf
[47] C. A. Cravotta and G. R. Watzlaf, “Design and performance of limestone drains to increase pH and remove metals from acidic mine drainage,” in Handbook of Groundwater Remediation using Permeable Reactive Barriers, Amsterdam, Netherlands: Elsevier, 2003, pp. 19–66. https://doi.org/10.1016/B978-012513563-4/50006-2
[48] E. Azzali et al., “Mineralogical and chemical variations of ochreous precipitates from acid sulphate waters (asw) at the Roşia Montană gold mine (Romania),” Environ. Earth Sci., vol. 72, no. 9, pp. 3567–3584, Nov. 2014. https://doi.org/10.1007/s12665-014-3264-z
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
Recibido: 10 de abril de 2025; Aceptado: 1 de septiembre de 2025
Abstract
Context:
The mining industry is the main culprit behind the generation of acid mine drainage (AMD). During coal extraction processes, sulfide minerals react with groundwater, releasing ions such as Fe2+ and Fe3+, sulfates (SO4 −2 ), and protonic acidity (H+). The low pH of AMD can cause significant environmental damage. AMD remediation is usually achieved using alkaline systems, wherein the AMD passes through limestone to be neutralized. Nevertheless, this process requires prolonged treatment times and constant cleaning steps to remove the coating formed on the limestone, which reduces its effectiveness.
Method:
This study evaluates a novel oxic-limestone rotational system for the treatment of AMD produced by the coal industry. The AMD collected was characterized in terms of pH, dissolved oxygen, Fe (Fe total, Fe2+, and Fe3+), and SO4 −2.
Results:
The results demonstrate the optimal efficiency of the proposed system, reducing the treatment time from 120 h in conventional systems to 1.5 h when applying a ratio of 0.25k g of limestone per liter of AMD.
Conclusions:
The rotational system enables the superficial degradation of the limestone, maintaining an active contact area for longer periods. This allows for optimized AMD remediation efficiency, reducing operating costs and necessitating fewer system cleanup steps.
Keywords:
coal mining, acid mine drainage, rotational system, superficial degradation.Resumen
Contexto:
La industria minera es el principal responsable de la generación de drenaje ácido minero (DAM). Durante los procesos de extracción del carbón, los minerales sulfurados reaccionan con las aguas subterráneas, liberando iones como Fe2+ y Fe3+, sulfatos (SO4 −2), y acidez protónica (H+). El bajo pH del DAM puede generar importantes daños medioambientales. La remediación del DAM generalmente se realiza a través de sistemas alcalinos, donde el DAM pasa a través de roca caliza para ser neutralizado. No obstante, este proceso requiere tiempos prolongados de tratamiento y etapas de limpieza constante para eliminar el recubrimiento formado en la roca, lo que reduce su eficacia.
Método:
Este estudio evalúa un novedoso sistema rotacional de óxido-caliza para el tratamiento de DAM producido por la industria del carbón. El DAM recolectado se caracterizó en términos de pH, oxígeno disuelto, Fe (Fe total, Fe2+ y Fe3+), y (SO4 −2).
Resultados:
Los resultados demuestran la eficiencia óptima del sistema propuesto, que reduce el tiempo de tratamiento de 120 h con sistemas convencionales a 1.5 h al aplicar una relación de 0.25 kg de caliza por litro de DAM.
Conclusiones:
El sistema rotacional facilita la degradación superficial de la caliza, manteniendo un área de contacto activa durante periodos más largos. Esto permite optimizar la eficiencia de remediación del DAM, reduciendo los costes de operación y las etapas de limpieza requeridas por el sistema.
Palabras clave:
minería del carbón, drenaje ácido minero, sistema rotacional, degradación superficial.Introduction
The acid mine drainage (AMD) produced during mining activities is a polluting byproduct formed by the reaction between minerals, oxygen, and water 1 . In the case of coal mining processes, AMD is characterized by a low pH and a high concentration of ions in the form of SO4 2-, Fe2+, and Fe3+, produced by the oxidation of pyrite (FeS2), in addition to other contaminating metal ions in lower proportions, such as Mn2+ and Al3+, which are typical of AMD waste 2)(5. The concentrations of these ions vary according to the geology of the area where the mining activity takes place 6.
In Colombia, the coal industry is one of the main mining activities, carried out mainly in the Boyacá region. This region contributes 38 % of the national coal production, which represents 3.087 million metric tons destined for use in the thermal and metallurgical sectors 7)(8. Coal mining operations in the area are not efficient, producing a high amount of pollutants that threaten nearby tributaries 9)(10. This problem is exacerbated by stormwater runoff, which can carry large amounts of AMD along with particulates and dissolved materials, substantially reducing the oxygen and nutrient content of the soil, as well as degrading aquatic systems and dependent biota 11)(12.
The application of systems such as aerobic wetlands and limestone drainage are potential alternatives that allow treating AMD 13)(14. The application of an active system (oxic) enables a higher rate of neutralization and removal of metal contaminants 15)(16. The high concentrations of SO4 2- depend on the mineralogy of the place 17)(18, which makes water treatment difficult. Furthermore, the efficiency of open limestone channel (OLC) systems is compromised by the formation of gypsum (CaSO4·2H2O) on the limestone surfaces, acting as a barrier that reduces its effectiveness during AMD treatment 19).
Similarly, optimal Fe removal values can be obtained using these systems 3. Nevertheless, these systems require continuous maintenance stages for their proper functioning, in addition to prolonged periods of fluid retention 20).
Considering the above, this study aims to evaluate the application of a novel rotational oxic limestone system to optimize AMD treatment processes. As a control mechanism, a non- moving device is used, called a static system. Tests were carried out in a controlled manner, using AMD samples collected from the area of interest. The authors did not find literature related to the evaluation of the effect of alkalization and the treatment of AMD using similar devices, which reinforces the novelty of this research.
Experimentation
The AMD used in this study was supplied by Cooperativa Agro-Minera (Coagromin Ltda.), located at Km 5 of the Paipa-Tunja road in Boyacá, Colombia. The AMD was later processed by adding limestone with a particle size between 1.27 and 2.54 cm. The limestone used was collected from Cantera Metrópolis, located at Km 6.6 of the Moniquirá-Arcabuco road (Boyacá). The limestone used in this study was analyzed via X-ray diffraction (XRD) in a Pananytical diffractometer (Co, λ = 1.75 Å). The analysis of the XRD pattern was carried out using the HighScore-Plus software, the Crystallography Open Database (COD), and the Inorganic Crystal Structure Database (ICSD).
Static tests
Static tests were performed on the system presented in Fig. 1. The proposed system was isolated during the analysis period in order to avoid agitation, favoring a greater sedimentation and settling of the compounds that may be generated. The tests were conducted under controlled conditions, i.e., at 2800 meters above sea level (m.a.s.l.), with a pressure of 740 hPa and an average temperature of 17 °C 21. The limestone:AMD ratio was 2.5:1 (in weight), ensuring the total coverage and reaction of the AMD. In this study, we evaluated the concentration of Fe ions (i.e., Fe2+ and Fe3+) and sulfates SO4 2- considering that these are the main dissolved ions in AMD from coal mining processes due to the presence of pyrite 22. Therefore, other trace metals in AMD samples were not characterized. The tests were carried out until an optimal removal of Fe (total, Fe2+, and Fe3+) and SO4 2- was achieved. Fe and sulfate measurements were taken using the FerroVer (iron reagent, Hach, USA) and SulfaVer IV (sulfate reagent, Hach, USA) reagents along with a DR6000 spectrophotometer (Hach, USA). The dissolved oxygen (DO) content was obtained from a Hach-Flexi HQ30d oximeter, and pH measurements were carried out using a SCOTT HandyLab pH11. Alkalinity was determined using 25 mL of undiluted AMD samples mixed with phenolphthalein and bromocresol green. The AMD sample was titrated with 0.02 N H2SO4 (i.e., 10 mol·m−3). Likewise, acidity was evaluated by adding phenolphthalein as an indicator solution. The mixture was also titrated with 0.02 N NaOH (i.e., 20 mol·m−3) 23.
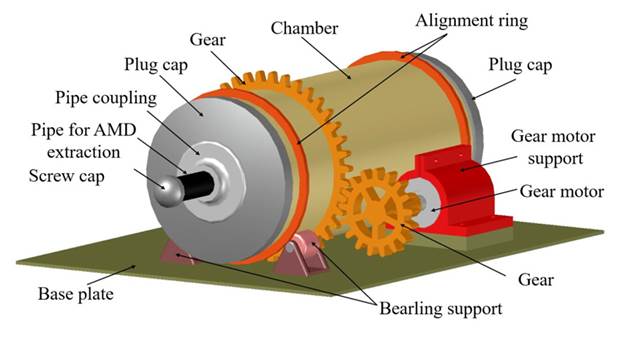
Figure 1: Schematic illustration of the AMD treatment device used in this study
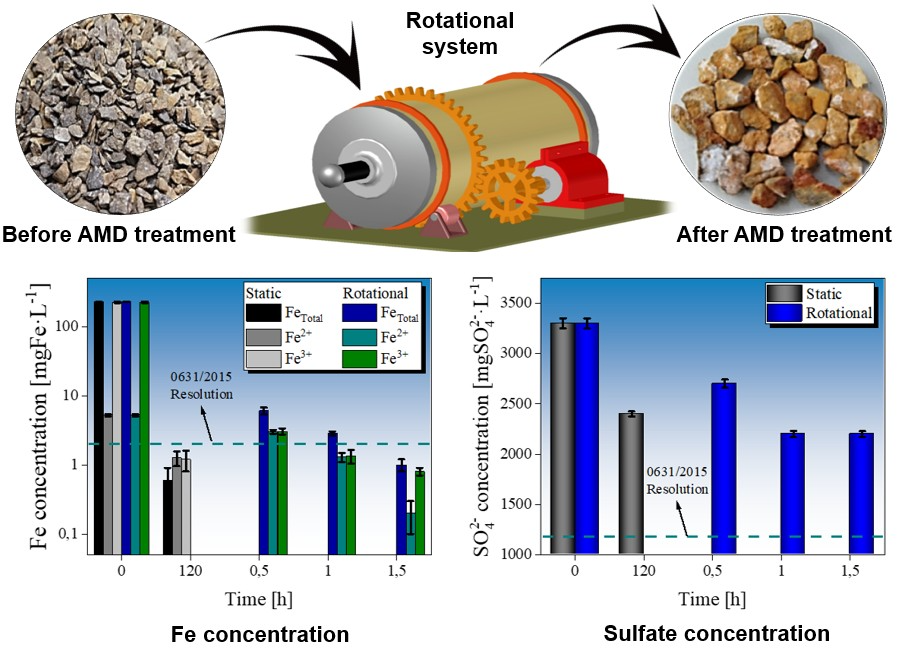
Dynamic tests using a rotational limestone system
Dynamic tests were performed in a two-reactor rotational mechanical system. The vessel had an internal length of 30 cm and a diameter of 12 cm, containing a maximum volume of 3393 cm3. The system was not operated at maximum capacity to preserve the functionality of the pressure gauges used to measure carbon dioxide (CO2). We employed a vessel inclination of 6° and a rotation speed of 11 rpm. Although an angle of 10° is typically used, as has been reported by 3)(24)(25, we opted for a smaller inclination to ensure a greater retention of the liquid within the system. Fig. 1 shows a scheme of the devices used in this study. 0.25 kg of limestone were used for every 1 L of AMD (Limestone:AMD ratio of 0.25:1). The mixing process was carried out for 0.5, 1, and 1.5 h. pH. DO, alkalinity, acidity, Fe2+, Fe3+, and SO4 2- measurements were made using the above-presented procedure.
Results
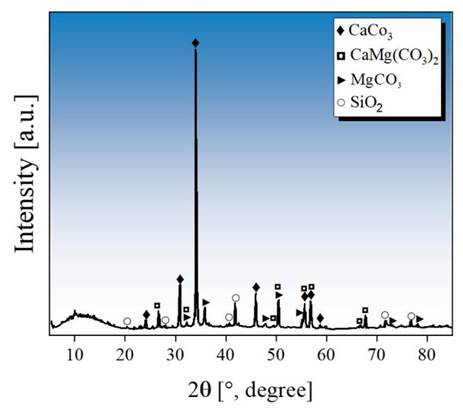
Fig. 2 shows the XRD pattern of the limestone used in this study. A semi-quantitative analysis, conducted using the Rietveld refinement method, revealed the presence of calcite—CaCO3, space group P 121/c1 14 , ICSD 150 26—, which was the main mineralogical compound (73.2 %). Likewise, we observed the presence of dolomite—9.3 %, CaMg(CO3)2, R¯3H (148), ICSD 10404 27—, magnesite—0.5 %, MgCO3, R¯3cH (167), ICSD 10264 28—, and silicates—17 %, mainly SiO2, P3221 (154), ICSD 16331 29—in a lower proportion. The composition of the limestone was similar to that found in other regions of Colombia, as reported by 30. A high content of carbonates in the limestone favors an optimal degree of alkalinity, as well as the limestone’s ability to react with AMD 31. The presence of amorphous material can be observed in the first degrees of diffraction, which corresponds to organic material typical of sedimentary rocks.
Figure 2: SXRD pattern of the limestone used in this study and collected in the region of Boyacá, Colombia
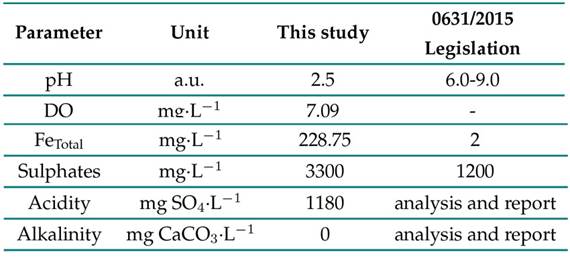
Figs. 3and 4 illustrate the behavior of the AMD when treated with a static and a rotational system, respectively. We followed the Colombian resolution no. 0631/2015 32 to determine the efficacy of both systems. An initial characterization of the AMD is presented in Table I. Note that the values are outside the permissible values as per the aforementioned resolution, which was issued by the Colombian Ministry of the Environment and Sustainable Development.
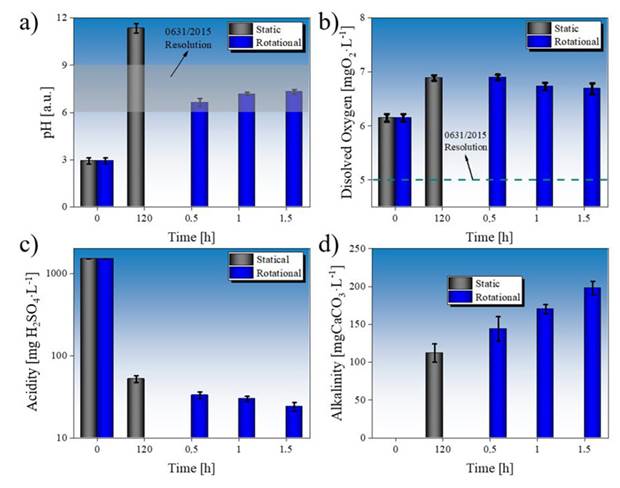
Figure 3: Values of a) pH, b) DO, c) acidity, and d) alkalinity for untreated and treated AMD in static and rotational systems. The pH and DO values obtained in this study were compared against those in resolution no. 0631/2015 for water quality 32
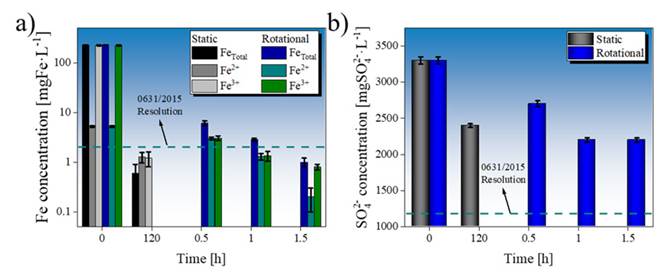
Figure 4: Concentration of a) Fe and b) sulfate ions in untreated and treated AMD after static and rotational treatment. The permissible values for pH and DO obtained in this study were compared against those in resolution no. 0631/2015 for water quality 32
Table I: Chemical characterization of the AMD sample used in this study and maximum permissible limit values for water resources linked to the extraction of coal and lignite according to Resolution 0631 of 2015 32
Discussion
Limestone is an inexpensive, natural, and efficient raw material for the treatment of AMD from coal mining processes. Dolomitic limestone, i.e., CaMg(CO3)2, is widely used within AMD systems due to its ease of reaction 33. Limestone reacts in the presence of hydrogen, enabling the release of carbonic acid, which can subsequently be converted into bicarbonate ions 3. The process is governed by the chemical reactions shown in Eqs. (1) and 2) (14)(34. This dissociation generates a buffer effect within the system that maintains stable pH values during the formation of metal precipitates. The presence of bicarbonate ions and limestone also favors the removal of Fe, according to Eqs. (3) and (4) 35) (36.
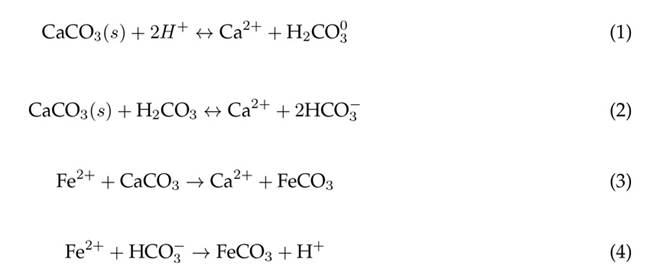
A lower rate of acidity reduction in AMD after rotational treatment for 0.5 h may be due to the release of acidity (H+) during the formation of siderite (FeCO3). Nevertheless, part of the H+ produced can also react with the limestone, which generates CO2, favoring carbonic acid formation 20)(36.

Although the generation of acidity by hydrogen should lower the pH of the solution, it can also be slowed down when carbonic acids come into contact with the limestone, as shown in Eq. (1), as well as by the formation of CO2 when reacting with part of the carbonic acids formed, i.e.,

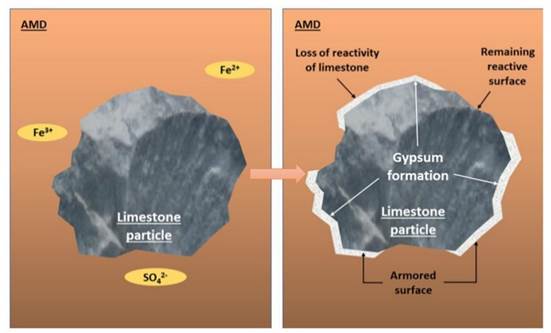
Likewise, the pH and alkalinity of the AMD can be leveled by the formation of passive layers of gypsum and siderite, which can be deposited on the surface of the limestone rock (Fig. 5). These precipitates act as a barrier that reduces the reactivity between the AMD and the rock 35)(37. Calcium sulfate (CaSO4) hydrates with two water molecules, forming gypsum (CaSO4·2H2O). Based on a stoichiometric analysis, for every 136.14 g of anhydrite, 36.03 g of water are required to form 172.17 g of gypsum. Therefore, no significant reduction in the volume of water in the system is expected due to this reaction. The volume of gypsum formed may be regarded as negligible, since these are very thin layers deposited on the limestone, formed to mitigate the rock’s activity.
Figure 5: Schematic illustration of gypsum binder formation from the reaction between AMD and limestone
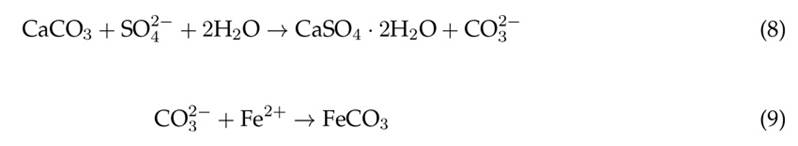
The formation of these passive layers allows explaining the low efficiency of the AMD treatment when occurring in a static system, which is unable to remove the precipitates deposited on the limestone surface 35)(38. Although the limestone:AMD ratio is ten times lower in the rotational system, the latter favors an autogenous grinding effect of the limestone 39, degrading the particles and removing the gypsum and siderite coatings.
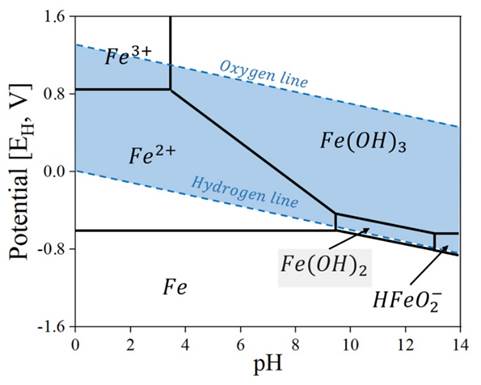
The delay in Fe2+ removal may be due to the low reactivity of this ion to generate compounds other than siderite within the AMD, so it is highly dependent on their reactivity with limestone. A DO concentration greater than 0.5 mg·L−1 favors the reaction of Fe2+ ions with AMD 35)(37)(40. The difficulties in creating other compounds, e.g., Fe(OH)2, which is greenish, is impaired by the formation of limonite, Fe(OH)3, from Fe3+, which has a 5/8 10YR (Munsell Soil Color Chart 41) color, as see in Fig. 6. Likewise, the formation of Fe(OH)3 is favored by pH values between 6.5 and 8 42)(45. Our measurement of the potential in the static and dynamic systems showed values between 0.1 and 0.15 V. As shown in Fig. 7, the final conditions obtained in this study favor the formation of the Fe(OH)3 mineralogical phase.
Figure 6: Photographs of the limestone samples used in this study a) before and b) after the AMD treatment. A change in the color of the limestone can be observed, which is due to the formation of precipitates on the rock surface
Figure 7: Generalized Pourbaix diagram for the Fe-H2O system. The pH values obtained in this work show agreater tendency towards the formation of the Fe(OH)3 phase
It has been reported that the Fe3+/Fe2+ ratio present in leachate from coal minerals can favor biodesulfurization through the reaction of sulfate ions, allowing for the formation of pyrite and, subsequently, the iron (II) (FeSO4) and iron (III) (Fe2(SO4)3) sulfate phases 20)(46. A reduction of Fe3+ ions, in addition to the potential formation of gypsum precipitates adhered to the limestone surface, explains the blockage during sulfate removal from the AMD. Stagnation in the process of removing sulfate ions could be observed. This is because the increase in pH caused by the interaction between the limestone and the AMD inhibits the reactive capacity of the limestone particles with the sulfate and iron (II) ions, so their concentrations are generally not significantly affected 47 . Although removal levels of 28 and 33 % were obtained for the samples treated in the static and rotational systems, respectively, it is necessary to apply secondary stages in order to comply with Colombian legislation. Even so, the application of a limestone system still exhibits an economic advantage compared to other technologies for the treatment of AMD and the removal of sulfates 35.
A system’s operation must be evaluated based on its working time, as well as on the cost of the raw materials. The application of our rotational system in this affected region is facilitated by the ease of obtaining limestone. The authors express their motivation for the further development of this project, which can help to address the effects generated by mining activities in the region. The application of raw materials from the affected area and the construction of passive treatment systems allows for significant cost reductions.
While these systems can be used in other regions and for different types of AMD, it is necessary to consider that the effectiveness of the treatment depends on the characteristics and initial conditions of the raw materials, i.e., the AMD and the limestone. In this study, the feasibility of this system was supported by the nearby availability of limestone. Therefore, when considering implementation in other affected areas, the costs of transporting limestone—as well as sustainability and logistical factors related to mining and transportation—must be considered.
During the AMD treatment, some residual materials were produced in the form of precipitates and clays. The chemical and toxic complexity of the waste generated currently precludes direct use in industrial activities due to the high risk of contamination associated with its release into the environment 48. The authors hope to conduct a feasibility analysis of the waste generated in subsequent studies, hoping to increase the sustainability of the proposal through new industrial products made from said waste.
Conclusion
This study evaluated the applicability of oxic-limestone drainage for the treatment of AMD and its effectiveness in the removal of Fe2+, Fe3+ and (SO4 2−)using a novel rotational system.
The application of a static system, which was used for comparison, required longer treatment times and yielded a lower ion removal efficiency compared to the rotational system. This was mainly due to the formation of precipitates that served as a barrier between the limestone and the AMD, hindering the system’s ability to react due to a reduction in the active area of the limestone. This barrier cannot be directly removed within the static system. In contrast, the dynamic behavior of our proposed solution generates an autogenous grinding process that favors the removal, by wear, of the precipitates deposited in the limestone, thus maintaining a continuous active contact area between the limestone and the AMD. The inability to remove sulfate ions was due to the premature depletion of Fe2+ ions and the formation of precipitates adhering to the limestone surface, which slowed the chemical reactions generated during the treatment of the AMD.
The authors would like to express their interest in the application of this type of system for the treatment of AMD and other leachates generated during different industrial activities in the region, as well as in the application of new raw materials from the area that allow for optimized industrial water treatment.
Acknowledgements
Acknowledgments
The authors are grateful to the Engineering Department of Universidad Pedagógica y Tecnológica de Colombia and Cooperativa Agrominera Multiactiva de Paipa Ltda. (Cooagromin, Paipa-Boyacá, Colombia) for providing the raw material and resources necessary for the proper development of this research. NRA would like to thank Conselho Nacional de Desenvolvimento Científico e Tecnológico do Brasil (CNPq, No. 408483/2022-9and No. 383215/2024-2) for the financial support provided.
References
Licencia
Derechos de autor 2025 Cesar René Blanco Zúñiga, Laura Daniela Ulloa-Amador, Nicolás Rojas-Arias

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-CompartirIgual 4.0.
A partir de la edición del V23N3 del año 2018 hacia adelante, se cambia la Licencia Creative Commons “Atribución—No Comercial – Sin Obra Derivada” a la siguiente:
Atribución - No Comercial – Compartir igual: esta licencia permite a otros distribuir, remezclar, retocar, y crear a partir de tu obra de modo no comercial, siempre y cuando te den crédito y licencien sus nuevas creaciones bajo las mismas condiciones.





2.jpg)