DOI:
https://doi.org/10.14483/23448393.21059Published:
2024-11-11Issue:
Vol. 29 No. 3 (2024): September-DecemberSection:
Chemical, Food, and Environmental EngineeringDesarrollo de una bebida alcohólica a partir de cubio (Tropaeolum tuberosum Ruiz & Pav.) empleando levadura de Saccharomyces bayanus
Development of an Alcoholic Beverage from Cubio (Tropaeolum tuberosum Ruiz & Pav.) Using Saccharomyces bayanus Yeast
Keywords:
bebida alcohólica, levadura, fermentación, tubérculo ancestral (es).Keywords:
alcoholic beverage, yeast, fermentation, ancestral tuber (en).Downloads
References
FAO, “Promover los cultivos marginados e infrautilizados,” 2017. [Online]. Available: http://www.fao.org/news/story/es/item/1032703/icode/
S. Sáenz-Torres, A. López-Molinello, L. Prieto, and T. Rodríguez, “El cubio como una alternativa productiva sostenible en condiciones de agricultura urbana de Bogotá,” Eq. Des., vol. 34, pp. 121-142, 2019. https://doi.org/10.19052/eq.vol1.iss34.6
R. Chirinos, H. Rogez, D. Campos, R. Pedreschi, and Y. Larondelle, “Optimization of extraction conditions of antioxidant phenolic compounds from mashua (Tropaeolum tuberosum Ruíz & Pavón) tubers,” Sep. Pur. Tech., vol. 55, pp. 217-225, 2007. https://doi.org/10.1016/j.seppur.2006.12.005
O. Ortega, D. Kliebenstein, C. Arbizu, R. Ortega, and C. Quiros, “Glucosinolate survey of cultivated and feral mashua (Tropaeolum tuberosum Ruíz & Pavón) in the Cuzco region of Perú,” Econ. Botany, vol. 60, no. 3, pp. 254-264, 2006. https://doi.org/10.1663/0013-0001(2006)60[254:GSOCAF]2.0.CO;2
F. M. Mejia, J. E. Salcedo, and S. Vargas, “Capacidad antioxidante y antimicrobiana de tubérculos andinos (Tropaeolum tuberosum y Ullucus tuberosus),” Rev. U.D.C.A., vol. 21 no. 2, pp. 449-456, 2018. https://doi.org/10.31910/rudca.v21.n2.2018.1083
P. Djabayan, L. González, M. Lucena, and C. Valarezo, “Aislamiento y actividad biológica de lectinas obtenidas de semillas de frutas, granos y tubérculos de plantas andinas,” Info. Tecnol., vol. 33, no. 2, pp. 21-36, 2022. https://doi.org/10.4067/S0718-07642022000200021
A. C. Morillo, Y. Morillo, and Y. P. Tovar, “Caracterización molecular de cubios (Tropaeolum tuberosum Ruíz y Pavón) en el departamento de Boyacá,” Rev. Cien. Agr., vol. 33, no. 2, pp. 32-42, 2016. http://dx.doi.org/10.22267/rcia.163302.50
S. E. Aguirre, N.V. Piraneque, and I. Pérez, “Sistema de producción de tubérculos andinos en Boyacá, Colombia,” Cuad. Des. Rural, vol. 9, no. 69, pp. 257-273, 2012.
M. B. Suquilanda-Valdivieso, “Producción orgánica de mashwa (Treopaeolum tubersum),” en Producción orgánica de cultivos Andinos (Manual Técnico), M. B. Suquilanda-Valdivieso, Ed., Ecuador: FAO, UNOCANC, Ministerio de Agricultura, Ganadería, Acuacultura y Pesca, 2012, ch. 2, pp. 19-31. http://www.fao.org/familyfarming/detail/en/c/384373/
G. Miller, “Use of dinitrosalicylic acid reagent for determination of reducing sugar,” Analytical Chem., vol. 31, pp. 426-428, 1959. https://doi.org/10.1021/ac60147a030
Density of sucrose solutions at 0-100°C and 0-70%, Official Method of Analysis 962.37, AOAC International, 1995.
Protein (crude) in animal feed, forage (plant tissue), grain, and oilseeds, Block digestion, Steam distillation method, Official Method of Analysis 2001.11, AOAC International, 2005.
Ash and fruit products, gravimetric method, alkalinity, Official Method of Analysis 940.26, AOAC International, 2005.
Y. Göksungur and U. Güvenç, “Enzymatic hydrolysis and production of ethanol from potato starch,” Gida, vol. 19, no. 2, pp. 89-92, 1994.
L. Sánchez and L. M. Triviño, “Obtención de alcohol etílico a partir de la fermentación de Saccharomyces bayanus, en un clon promisorio de papa criolla en (Solanum tuberosum Grupo Phureja) cosechado en el Municipio de El Rosal,” undergraduate thesis, Escuela de Ciencias Básicas, Tecnología e Ingeniería, Universidad Nacional Abierta y a Distancia, Colombia, 2014.
A. Ronquillo, R. Lazcano, I. Pérez, S. Cabrera, and S. Lazcano, “Elaboración y caracterización de vino de frutas e infusión de hierbas,” Inv. Des. Cien. Tecnol. Alim., vol. 1, no. 1, pp. 366-371, 2016.
Bebidas alcohólicas. Métodos para determinar el contenido de alcohol, Norma Técnica Colombiana NTC 5113, ICONTEC Internacional, Colombia, 2003.
Bebidas alcohólicas. Métodos para determinar la acidez y el pH, Norma Técnica Colombiana NTC 5114, ICONTEC Internacional, Colombia, 2003.
Bebidas alcohólicas. Método colorimétrico para determinar el contenido de metanol, Norma Técnica Colombiana NTC 5159, ICONTEC Internacional, Colombia, 2003.
Bebidas alcohólicas. Método para determinar el extracto, Norma Técnica Colombiana NTC 5162, ICONTEC Internacional, Colombia, 2003.
Colorimetry-Part 6: CIEDE 2000-Colour difference formula, ISO/CIE 11664-6:2014, International Organization for Standardization ISO, 2014.
Bebidas alcohólicas. Vinos de frutas, Norma Técnica Colombiana NTC 708, ICONTEC Internacional, Colombia, 2000.
Statistix 10. Data analysis software for researchers. (2013). Analytical Software.
S. Sáenz-Torres, L. Prieto, and A. López-Molinello, El cubio (Mashua): producción, transformación y beneficios, Bogotá, Colombia: Ediciones Unisalle, 2020. https://doi.org/10.19052/9789585148413
B. Aguado, “Influencia del tiempo de soleado sobre la concentración de azúcares reductores y capacidad antioxidante de la mashua (Tropaeolum tuberosum) ecotipo negra,” undergraduate thesis, Escuela Profesional de Ingeniería Agroindustrial, Universidad Nacional de Huancavelica, Perú, 2017.
D. Arteaga, L. Chacón, V. Samame, D. Valverde, and L. M. Paucar, “Mashua (Tropaeolum tuberosum): composición nutricional, características químicas, compuestos bioactivos y propiedades beneficiosas para la salud,” Agroind. Sci., vol. 12, no. 1, pp. 95-101, 2022. https://doi.org/10.17268/agroind.sci.2022.01.12
D. Romero and L. Tuirán, “Caracterización fisicoquímica, funcional, reológica y composicional de la harina precocida de cubio (Tropaeolum tuberosum R&P) cultivado en diferentes fuentes de fertilización,” undergraduate thesis, Department of Engineering, Universidad de La Salle, Colombia, 2017.
F. Velásquez, E. Ramirez, R. Chuquilin, and I. Aliaga, “Optimization of the functional properties of a drink based on tubers of purple mashua (Tropaeolum tuberosum Ruíz y Pavón,” Agroind. Sci., vol. 10, no.1, pp. 63-70, 2020. https://doi.org/10.17268/agroind.sci.2020.01.09
A. Castañeda-Sánchez and J. A. Guerrero-Beltrán, “Pigmentos en frutas y hortalizas rojas: antocianinas,” Temas Sel. INg. Alim., vol. 9, no. 1, pp. 25-33, 2015.
Y. Zhao, S. Li, Q. Shu, X. Yang, and Y. Deng, “Highly efficient production of 2-phenylethanol by wild-type Saccharomyces bayanus strain,” Biores. Tech., vol. 403, no. 1, pp. 1-10, 2024. https://doi.org/10.1016/j.biortech.2024.130867
H. Kretzschma, Levaduras y alcoholes y otros productos de la fermentación, Barcelona, Spain: Editorial Reverté, 1998.
I. S. Pretorius, “Tailoring wine yeast for the new millennium: Novel approaches to the ancient art of winemaking,” Yeast, vol. 15, pp. 675-629, 2000. https://doi.org/10.1002/1097-0061(20000615)16:8<675::AID-YEA585>3.0.CO;2-B
M. Bonatto, M. Gonçalves, A. C. Conti-Silva, and V. Luiz, “Influence of two different vinification procedures on the physicochemical and sensory properties of Brazilian non-Vitis vinifera red wines,” LWT Food Sci. Tech., vol. 54, no. 2. pp. 360-366, 2013. https://doi.org/10.1016/j.lwt.2013.06.020
C. Granados, M. E. Torrenegra, D. Acevedo, and P. Romero, “Evaluación fisicoquímica y microbiológica del aperitivo vínico de lulo (Solanum quitoense L.),” Información tecnológica, vol. 24, no. 6, pp. 35-40, 2013. https://dx.doi.org/10.4067/S0718-07642013000600006
D. Huang, Y. Zhong, Y. Liu, Y. Song, X. Zhao, and Y. Qin, “Reducing higher alcohols by integrating indigenous Saccharomyces cerevisiae, nitrogen compensation, and chaptalization methods during fermentation of kiwifruit wine,” LWT, vol. 184, p. 115059, 2023, doi: https://doi.org/10.1016/j.lwt.2023.115059
S. Bian et al., “Effects of the addition of thermostable α-amylase on the physicochemical and antioxidant properties of extrusion-pretreated Apios fortunei used for yellow wine fermentation,” LWT, vol. 154, p. 112845, 2022. https://doi.org/10.1016/j.lwt.2021.112845
E. Kolb, Vinos de frutas elaboración artesanal e industrial, Zaragoza, Spain: Editorial Acribia S.A., 2002.
D. Ming-Yu, H. Koizumi, and Y. Suzuki, “Comparison of three chromatographic systems for determination of organic acids in wine,” Analytical Sci.. vol. 11, no. 2, pp. 239-243, 1995. https://doi.org/10.2116/analsci.11.239
A. Pastore, D. Badocco, L. Cappellin, M. Tubiana, and P. Pastore, “Real-time monitoring of the pH of white wine and beer with colorimetric sensor arrays (CSAs),” Food Chem., vol. 452, p. 139513, 2024, doi: https://doi.org/10.1016/j.foodchem.2024.139513
N. Khadka et al., “Study on the changes during the fermentation of the wine prepared from palm (Phoenix sylvestris) sap,” Heliyon, vol. 10, no. 15, p. e35799, 2024. https://doi.org/10.1016/j.heliyon.2024.e35799
L. García, M. Flórez, C. Marrugo, and Y. Ligardo, “Elaboración y caracterización fisicoquímica de un vino joven de fruta de borojo (B patinoi Cuatrec),” Cien. Doc. Tecnol., vol. 27, no. 52, pp. 507-519, 2016.
L. M. Quispe, “Obtención de una bebida alcohólica a partir de la fermentación de leche,” Rev. Inv. Cien. UNTRM: Cien. Nat. Ing., vol. 1, no. 3, pp. 46-51, 2018. https://doi.org/10.25127/ucni.v1i3.425
R. Salazar, G. Espinoza, C. Ruiz, M. F. Fernández, and R. Rojas, “Compuestos fenólicos, actividad antioxidante, contenido de resveratrol y componentes del aroma de 8 vinos peruanos,” Rev. Soc. Quím. Perú, vol. 77, no. 2, pp. 135-143, 2011.
S. V. Olivares, M. Silva, and R. A. Caruajulca, “Bebida alcohólica por fermentación de cáscara y mucilago del café (Coffea arabica L.) a diferente pH y concentración de levadura,” Rev. Inv. Cien. UNTRM: Cien. Nat. Ing., vol. 3, no. 1, pp. 9-15, 2020. http://dx.doi.org/10.25127/ucni.v3i1.586
F. J. Francis and P. C. Markakis, “Food colorants: Anthocyanins,” Crit. Rev. Food Sci. Nut., vol. 28, no. 4, pp. 273-314, 1989. https://doi.org/10.1080/10408398909527503
J. Liu et al., “Ethyl esters enhancement of Jinchuan pear wine studied by coculturing Saccharomyces bayanus with Torulaspora delbrueckii and their community and interaction characteristics,” Food Biosci., vol. 46, art. 101605, 2022. https://doi.org/10.1016/j.fbio.2022.101605
P. Ribéreau, D. Dubourdieu, B. Donèche, and A. Lonvaud, Handbook of enology: The microbiology of wine and vinifications, London, UK: John Wiley & Sons, 2006.
S. J. Bloor and R. Falshaw, “Covalently linked anthocyanin-flavonol pigments from blue Agapanthus flowers,” Phytochem., vol. 53, no. 5, pp. 575-579, 2000. https://doi.org/10.1016/S0031-9422(99)00572-5
I. Povea, La función del envase en la conservación de alimentos, Bogotá, Colombia: ECOE Editores, 2014.
How to Cite
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Download Citation
Recibido: 24 de julio de 2023; Aceptado: 20 de agosto de 2024
Abstract
Context:
Cubio (Tropaeolum tuberosum), an ancestral tuber cultivated in select regions of Colombia, possesses significant nutritional value but remains a marginalized crop. This study aims to enhance the recognition of cubio by diversifying its applications through the production of a fermented alcoholic beverage.
Method:
Initially, the cubio tuber was characterized. A concentrated and enzymatic pretreatment utilizing alpha-amylase and glucoamylase was employed to release sugars. Afterwards, fermentation was carried out, using Saccharomyces bayanus in a carefully controlled bioreactor setting. The fermentation medium was composed of 0.1 % (NH4) H2PO4, 0.3 % MgSO4, 0.5 % peptone, and 0.3 % yeast extract, with the aim of reaching an alcohol content of 15 °GL. The bioreactor was inoculated with Saccharomyces bayanus at 25 °C, maintaining a pH of 5.2 and an agitation speed of 100 rpm. Daily monitoring of pH levels, sugar concentrations, and biomass was conducted over a 15-day period. Finally, the physicochemical properties, physical attributes, and sensory qualities of the resulting product were assessed.
Results:
The analysis of the cubio tuber revealed the presence of 2.18 ± 0.002 % reducing sugars, 4.48 ± 0.064 % proteins, and 1.66 ± 0.015 % starch. Throughout the fermentation process, the yeast exhibited a growth rate of 2.3786 mg.L−1.day−1. As a result, the concentration of reducing sugars decreased to 15.45 mg.L−1.day−1, and an alcohol production of 1.1786 ºGL.day−1 was observed. The final alcoholic beverage contained 15.24 °GL, with a pH of 4.5 and a total acidity of 7.21 ± 0.029 g. L−1. Additionally, an expert panel positively rated the flavor and aroma of the beverage.
Conclusions:
It is technologically possible to produce a fermented alcoholic beverage of adequate quality from the cubio tuber, implying an alternative use for this ancestral crop.
Keywords:
alcoholic beverage, yeast, fermentation, ancestral tuber.Resumen
Contexto:
Cubio (Tropaeolum tuberosum), un tubérculo ancestral cultivado en regiones selectas de Colombia, posee valor nutricional significante pero continúa siendo marginalizado. Este estudio busca expandir el reconocimiento del cubio al diversificar sus aplicaciones mediante la producción de una bebida alcohólica fermentada.
Método:
Inicialmente, el tubérculo de cubio fue caracterizado. Se empleó un pretratamiento concentrado y enzimático para liberar azúcares. Después se llevó a cabo el proceso de fermentación, utilizando Saccharomyces bayanus en un biorreactor cuidadosamente configurado y controlado. El medio de la fermentación estaba compuesto de 0.1 % de (NH4) H2PO4, 0.3 % de MgSO4, 0.5 % de peptona y 0.3 % de extracto de levadura, a fin de alcanzar un contenido de alcohol de 15 °GL. El biorreactor fue inoculado con Saccharomyces bayanus a 25 °C, manteniendo un pH de 5.2 y una velocidad de agitación de 100 rpm. Se monitorearon diariamente los niveles de pH, la concentración de azúcar y la biomasa a lo largo de un periodo de 15 días. Finalmente, se evaluaron las propiedades fisicoquímicas, los atributos físicos y las cualidades sensoriales del producto resultante.
Resultados:
El análisis del tubérculo de cubio evidenció la presencia de 2.18 ± 0.002 % de azúcares reductores, 4.48 ± 0.064 % de proteína y 1.66 ± 0.015 % de almidón. A lo largo del proceso de fermentación, la levadura mostró una tasa de crecimiento de 2.3786 mg.L−1.día−1. Como resultado, la concentración de azúcares reductores se redujo a 15.45 mg.L−1.día−1, y se observó una producción de alcohol de 1.1786 ºGL.día−1. La bebida alcohólica final contenía 15.24 °GL, con un pH de 4.5 y una acidez total de 7.21 ± 0.029 g. L−1. Adicionalmente, un panel de expertos calificó positivamente el sabor y el aroma de la bebida.
Conclusiones:
Es tecnológicamente posible producir una bebida fermentada alcohólica de calidad adecuada a partir de tubérculo de cubio, lo que implica un uso alternativo para este cultivo ancestral.
Palabras clave:
bebida alcohólica, levadura, fermentación, tubérculo ancestral.Introduction
In recent years, there has been growing interest in reevaluating and rediscovering ancient foods for their nutritional value, flavors, unique characteristics, and textures. The Food and Agriculture Organization of the United Nations (FAO) has been urging its member countries to explore the technological potential of alternatives categorized as underutilized and neglected crops, secondary crops, or promising crops. Colombia has been proactive in this commitment, leading to the enactment of Law 2046 of 2020 by Congress, which promotes the cultivation, consumption, and technological use of ancestral crops. Among these crops, certain tubers have been preserved with the help of indigenous communities and farmers, one of them being the tuber known as cubio (Tropaeolum tuberosum Ruiz & Pav. 1802, Brassicales: Tropaeolaceae), which is native to the South American Andes. However, as emphasized by the FAO 1, the limited allocation of resources by governments for the promotion and development of these species has led to missed opportunities. Highlighting the need for political will, this research underscores the potential to promote the consumption and processing of cubio through the production of a distilled beverage as a technological alternative that meets the established parameters for such products 2.
Cubio is known by various names, such as añu, apiñu, cubio, isañu, isaña, isaño, pañí, puel, majua, mauja, mashwa, or mashua, depending on the Andean region where it is cultivated. It has lower commercialization compared to other Andean crops such as potato (Solanum tuberosum L.), oca (Oxalis tuberosa), and ulluco (Ullucus tuberosus), among others. One reason for this is that cubio is associated with a bitter taste, which is caused by the presence of isothiocyanates. However, these bitter compounds also serve as important phenolic antioxidants, with potential health benefits (3-5). This is why cubio is attributed antimicrobial 5,6, hemagglutinating, and anticoagulant properties 6.
Colombian cubio exhibits a range of skin colors, from white to dark violet-purple, with additional shades of yellow, orange, red, and pink. Despite its diversity and historical significance as a food source for ancient civilizations, cubio, along with other Andean tubers and roots such as ibia, rubas, and yacón, faces the risk of genetic erosion 7. This poses a threat to their continued existence in the territory. However, efforts to empower low-income communities through the technical knowledge and production management of these species offer a potential solution. Simple and cost-effective processes can transform these tubers into flours, beverages, nectars, jams, sweets, and bakery products, expanding their utilization and market opportunities, particularly among the younger generations, who have reduced their consumption 2.
The effort to contribute to the prevention of cubio erosion - note that this tuber is cultivated in undemanding soils - inspires this research to offer alternative products to those traditionally made with it, thereby preserving and expanding the market for alcoholic beverages 8, e.g., in the form of a wine that meets people’s expectations 9.
Considering the above, the objective of the research was to evaluate a beverage obtained through the alcoholic fermentation of the cubio tuber (Tropaeolum tuberosum R&P), specifically focusing on the Colombian variety known as blanca ojo morado (white purple-eyed). Winemaking techniques were applied, utilizing wine yeasts, in order to explore cubio as a potential raw material for alcoholic beverage production, adapting the practices used in grape wine production.
This study holds significance regarding the preservation and promotion of the use of cubio, a valuable yet underutilized tuber, while simultaneously offering an alternative product of good nutritional and sensory quality. By bridging traditional knowledge with modern fermentation practices, our proposal seeks to contribute to the sustainable utilization and commercialization of cubio, thereby benefitting local communities and safeguarding the genetic diversity of these important crops.
Materials and methods
Physicochemical characterization of the cubio tuber (blanca ojo morado variety)
In this study, three samples of cubio were analyzed in triplicate, aiming to determine the carbon and nitrogen content present in the tuber, which is utilized by the yeast during fermentation. As a carbon source, the tubers’ sugars and starch contents were measured through Miller’s acid hydrolysis method 10 and the procedure established in AOAC International’s 985.29 method 11. Additionally, its dry matter and moisture were determined via the gravimetric method described in 12, and the protein content (nitrogen source) was measured using AOAC International’s 2001.11 method 13.
Conditioning the cubio tuber
The process started by subjecting 2 kg of cubio to immersion washing, effectively removing any external impurities. To ensure proper disinfection, a solution of chlorinated alkyl dimethyl benzyl ammonium was prepared with a ratio of 1:10 (disinfectant:water), in order to treat the cubio tuber.
To facilitate the interaction between cubio nutrients and the yeast during fermentation, the tuber was liquefied. The liquefaction process aimed to break down the tuber, allowing for a better nutrient release. The filtered solids from this extract were washed across several stages, ensuring that the maximum amount of fermentable material was available. This concentration step was crucial in optimizing the fermentation process, as it provided the yeast with a richer source of sugars to convert into alcohol, thereby enhancing the efficiency and yield of the overall process. The resulting liquefied cubio was then adjusted to a volume of 2 L to prepare the must, which would serve as the fermentation medium. To further enhance the characteristics of the must, it was heated to 60 °C in a kettle. This heating step helped to fix the color of the cubio - this process potentially influences other flavor and aroma components. Following the heating process, the must was cooled down to an appropriate temperature and subsequently transferred to a BioFlo 110 bioreactor, which provided a controlled environment for the fermentation process, enabling the precise monitoring and regulation of various parameters.
By means of these specific procedures, this study aimed to optimize the preparation of cubio must for saccharification and fermentation while ensuring the desired final characteristics and qualities throughout the process.
Preparing the must for fermentation
For the subsequent steps of the fermentation process, we followed the protocol developed by 14, as modified by 15. The 2 L of liquefied and concentrated cubio in the bioreactor underwent pre-liquefaction and post-liquefaction, each performed under specific temperature and pH conditions.
During the pre-liquefaction stage, the liquefied cubio was subjected to a temperature of 70 °C and a pH of 7.0 for 60 min. This step helped to further break down the tuber components and prepare them for subsequent processes. Additionally, the enzyme α-amylase BAN 480L (50 g. hL-1 concentration) was added to facilitate the liquefaction process.
During the post-liquefaction stage, the liquefied cubio was maintained at a temperature of 70 °C and a pH of 5.2 for 90 min. The modified conditions of this stage helped to optimize the breakdown of starch and other complex carbohydrates present in the cubio tuber. The addition of the enzyme α-amylase BAN 480L at a concentration of 50 g. hL−1 continued to support the liquefaction process.
After the liquefaction stages, saccharification was carried out. This process was conducted at a temperature of 65 °C and a pH of 5.2. The enzyme used for saccharification was the glucoamylase enzyme Amylase AG 300L, at a concentration of 0.4 %. This glucoamylase enzyme aids in the breakdown of complex sugars such as starch into simpler, fermentable ones, providing a suitable substrate for yeast fermentation.
By following this protocol, this study aimed to optimize the liquefaction and saccharification processes of the cubio tuber, ensuring the efficient conversion of complex carbohydrates into fermentable sugars for subsequent fermentation by the yeast.
Alcoholic fermentation
For the fermentation process, several nutrients were added to the must in order to support yeast growth and fermentation. These nutrients included 0.1 % (NH4) H2PO4, 0.3 % MgSO4, 0.5 % peptone, and 0.3 % yeast extract. These ingredients provided the essential elements and necessary compounds for yeast metabolism and fermentation. Additionally, Saccharomyces bayanus yeast (Safale US-05) 14 was introduced into the must to initiate fermentation.
The process was carried out for 15 days at a temperature of 24 °C in a fermentation vessel maintained at 100 rpm, ensuring adequate mixing and contact between the yeast and the must. The final pH of the fermentation medium was maintained at 4.5, creating favorable conditions for yeast activity.
For the fermentation process, we measured the content of reducing sugars derived from the cubio tuber, from the concentrated extract, and from the enzymatic breakdown of starch. The monitoring process helped to assess the progress of sugar consumption by the yeast during fermentation. Furthermore, additional sugar was added to the must in order to achieve the desired alcohol content of 15 °GL (Gay-Lussac). It was determined that 17 g of sugar produces 1° of alcohol 16. Sugar was added to reach 255 g and achieve the desired alcohol content. Daily observations and measurements were recorded during the fermentation process. These included data on reducing sugars (10), biomass (determined through constant drying) 12, pH levels, and alcohol content (°GL). The collected data provided valuable insights into the fermentation behavior and the progress of sugar utilization by the yeast over time. Graphical representations of the data were created to facilitate the analysis and interpretation of the fermentation dynamics.
Upon completion of the fermentation process, the alcoholic beverage was decanted to separate it from any sediment or solids. It was then subjected to filtration to further clarify the beverage and remove any remaining particles. After filtration, the fermented beverage was bottled and stored for subsequent evaluation and analysis.
The above-presented protocol enabled the controlled fermentation of the cubio must, resulting in the production of an alcoholic beverage. The recorded data and subsequent analysis provided valuable information on the fermentation kinetics, alcohol production, and the overall quality of the final product.
Characterization of the obtained alcoholic beverage
To assess the physicochemical properties of the fermented beverage, three samples were taken, and each property was analyzed in triplicate to ensure the accuracy and reliability of the measurements.
The following physicochemical properties were evaluated to verify compliance with the requirements of the Colombian Technical Standard NTC 708 for fruit wines, given that there is no national standard for this type of fermented beverages 22:
-
Alcohol content17. The alcohol content of the fermented beverage was determined using a specific method to quantify the percentage of alcohol present. This analysis provided crucial information on the strength of the beverage.
-
pH and total and volatile acidity18. The pH level and the total and volatile acidity of the beverage were measured. These parameters helped to assess the acidity and balance of the final product.
-
Methanol content19. Methanol, a toxic alcohol, was analyzed to ensure that its concentration in the fermented beverage adhered to safe levels for consumption.
-
Total dry extract20. We measured the total dry extract, which represents the concentration of solids in the beverage after removing the water. This parameter gave insights into the overall composition and density of the beverage.
In addition to the physicochemical properties, certain physical characteristics of the beverage were examined, i.e.,
-
Color21. The color of the beverage was evaluated using the Hunter method and a Konica Minolta colorimeter. This method measured color by analyzing Lab* or CIELAB spatial coordinates, and it provided information on the visual appearance and hue of the beverage.
-
Density11. We measured the density of the beverage, which relates to its mass per unit volume. This parameter gave insights into the concentration and overall texture of the beverage.
The collected data on physical and physicochemical characteristics were subjected to statistical analysis using the Statistix software, version 10.0 23. A completely randomized analysis of variance (ANOVA) was performed to determine significant differences between the samples. If significant differences between specific characteristics were found (p≤0.05), Tukey’s variance comparison test (p≤0.05) was applied for further analysis.
This comprehensive analysis allowed for a thorough evaluation of the fermented beverage, ensuring compliance with quality standards and providing insights into its physical and chemical properties.
Sensory evaluation
The flavor, color, and aroma attributes of the cubio wine were evaluated in a hedonic acceptance test with five expert wine tasting panelists. The obtained results were analyzed statistically using the Statistix software, version 10 23. Pearson’s correlation coefficient was employed to assess the correlations between the attributes (p≤0.05).
Results and discussion
Physicochemical characterization of cubio
The blanca ojo morado variety of cubio exhibited an average reducing sugars content of 2.18 ± 0.002 %. This aligns closely with the findings of 24, who reported a range of 2.16-2.83 % for the same variety. The author of 25, as cited by 26, reported a significantly higher value (7.2±0.24 %) for reducing sugars in the Peruvian cubio variety known as zapallo amarillo, emphasizing the potential differences between cubio varieties, which may also be influenced by variations in soil quality and the agricultural practices used during cultivation.
Regarding the starch content, the tubers exhibited an average value of 1.66 ± 0.015 %. This closely aligns with the value reported by 27 for the same cubio variety, which was 2.20 %. The presence of starch contributed to fermentation through enzymatic synthesis, providing additional reducing sugars as a substrate for yeast in the must.
As for the protein content, the cubio tubers exhibited an average value of 4.48 ± 0.064 %. This value was higher than that reported by 28 for purple cubio tubers (1.2±0.041 %). However, it was comparable to the value observed by 27 for the same variety (5.86 %) in a crop without fertilization.
The cubio tubers also exhibited an average dry matter content of 14.08 ± 0.002 %, contributing with solids to the fermentation process, including reducing sugars from starch as a carbon source and proteins as a nitrogen source for the metabolic development of yeast in the must. This finding aligns closely with those recorded by 24 for the blanca ojo morado variety cultivated with conventional fertilization, with 14.73 % dry matter and 85.27 % water. To preserve the anthocyanins or pigments in the cubio tubers, conditioning activities were promptly conducted to minimize degradation 29.
Alcoholic fermentation of cubio
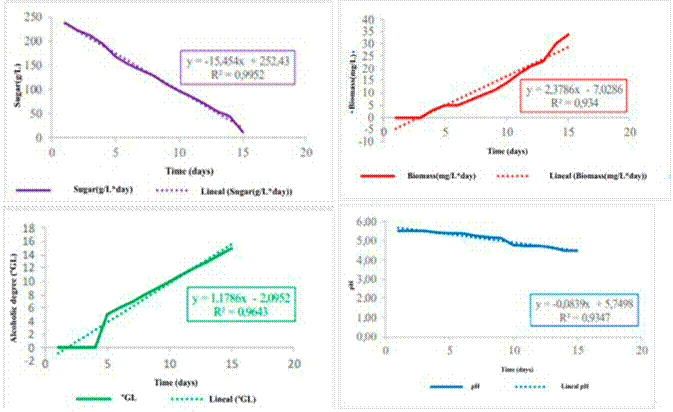
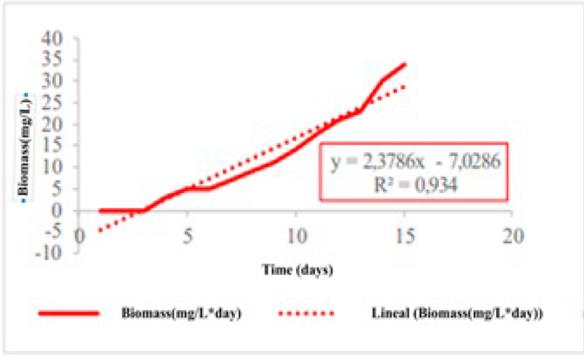
Throughout the course of the alcoholic fermentation process, parameters such as reducing sugars, biomass, alcohol generation, and pH in the must exhibited consistent trends, as illustrated in Fig. 1.
These trends showed linear variations over the 15-day fermentation period.
Figure 1: Alcoholic fermentation variables for the cubio beverage
Specifically, the Saccharomyces bayanus yeast employed in the fermentation process exhibited a growth rate of 2.3786 mg.L−1.day−1, given its ability to act from low-acid musts. This type of yeast can synthesize malic and succinic acid, inhibit malolactic fermentation, produce more glycerol than Saccharomyces cerevisiae, and generate less acetic acid 30, which indicates its capacity for multiplication and proliferation within the fermentation medium. Simultaneously, the concentration of reducing sugars experienced a gradual decrease, at a rate of 15.45 mg.L−1.day−1. This reduction can be attributed to the metabolic activity of the yeast, which utilizes the sugars as a carbon source for energy and growth.
Consequently, the fermentation process led to the production of alcohol at a rate of 1.1786 °GL.day−1. This steady production contributed to the gradual increase in the alcohol content of the beverage over the 15-day fermentation period. By the end of the process, the alcoholic beverage had reached a final alcohol content of 15.24 °GL.
These findings highlight the dynamic nature of fermentation, with the yeast exhibiting growth, sugar consumption, and alcohol production over time. The observed trends provide valuable insights into the kinetics and outcomes of the process.
The taste and aroma of alcoholic beverages are mainly influenced by alcohol and organic compounds present in smaller quantities 31. In our work, the pH and density of the must showed changes over time, indicating that alcoholic fermentation was taking place due to yeast metabolism.
The pH decreased at a rate of 0.0839 day−1, and the density reached a value of 0.9947 ± 0.002 g.cm−3.
Similar values were reported by 15 for alcoholic fermentation using fresh Criolla potato tubers and Saccharomyces bayanus yeast, with a biomass growth of 0.344 mg.mL−1.h−1, an increase of 1.8046 °GL.h−1 in alcohol content, and a decrease of 0.0045 mg.mL−1.h−1 in reducing sugars, demonstrating a similar behavior of yeast in fermentations with other tubers.
Before racking, the alcoholic beverage showed sediment formation at a rate of 0.4657 mL.h−1. This sedimentation confirmed that the yeast was actively transforming the components of the must through its fermentative power, degrading the sugars. The availability of nutrients in sediments during alcoholic beverage formation was confirmed by 32, including assimilable nitrogen, oxygen, vitamins, minerals, and ergosterol, as well as the presence of inhibitory substances.
Characterization of the alcoholic beverage obtained
The beverage analyzed in this study exhibited an alcohol concentration of 15.24 °GL, corresponding to 15.24 mL of ethanol per 100 mL of beverage. This value is close to that reported by 33, i.e., ranging from 8.6 to 14 °GL for Brazilian grape wines; to those observed by 34, with 11.3 °GL for a lulo wine; and to those recorded by 35 for fermented kiwifruit beverages, with 11.8 °GL. Alcohol content is important because it affects organoleptic characteristics such as the aroma and body of the wine 33. The saccharification stage with α-amylase not only aided in alcohol production by reducing the concentrated sugars derived from cubio (plus the sucrose added to the process) during fermentation; it also helped to increase the degree of gelatinization and the water solubility index, as well as to reduce the water absorption index and the viscosity of tuber extracts used in wine production 36. According to 37, fermented alcoholic beverages such as wine, beer, or cider should not exceed alcohol values of 17 g.100 g−1 in order to maintain desirable quality characteristics and a pleasant taste. This observation validates the potential of the cubio tuber as a new ingredient for alcoholic beverage production.
The total acidity was 7.21 ± 0.029 g.L−1, expressed as tartaric acid, which falls within the recommended range of 3.5 to 10 g.L−1 for wine-like alcoholic beverages, indicating the favorable quality achieved through controlled fermentation 38. The obtained value falls within the range reported by 36, i.e., between 5.45 and 8.4 g.L−1 for tuber wines. When the total acidity expressed as tartaric acid is above 2 g.L−1, it further supports beverage quality. Indeed, pH and acidity are closely related to the solubility of tartaric salts 39. Regarding the total acidity, 40 reported a value of 4.2 g.L−1 when using a Saccharomyces cerevisiae strain (to induce fermentation) and a different substrate such as palm (Phoenix sylvestris) sap. Their result was lower than ours. Diverse strains and substrates may influence the accumulation of total acidity (as tartaric acid).
The average methanol content in the beverage was 1.0327 ± 0.00079 mg.L−1, indicating fermentation stability and quality. The presence of trace amounts of methanol confirms that the process was primarily focused on ethanol production, resulting in a beverage that is safe and suitable for consumption. The obtained value is below the maximum limit specified in the NTC 708 standard (<1000 mg.dm−3 of anhydrous alcohol) 22. It is important to mention that surpassing this limit, as noted by 41, can result in metabolic intoxication. Therefore, given its neurotoxic effects, methanol is an unwanted component in any kind of wines 35.
The density of the beverage was 0.9947 ± 0.002 mg.mL−1, indicating the presence of alcohol resulting from yeast activation during the fermentation process. This density value exhibits a direct relationship with the concentration of sugars in the beverage. Similar results were obtained by 42 in the production of a non-distilled alcoholic beverage from milk, where a density value of 1.034 was reported. Furthermore, 40) reported values of 0.99 mg. mL−1, and 43 recorded 0.9916 and 1.0174 mg. mL−1 for Peruvian grape wines.
The pH of the beverage was 4.585 ± 0.0018, which falls within the optimal range and is similar to that obtained by 40 (4.2) and by 44 (4.5) during the fermentation of palm sap and coffee husk and mucilage, respectively, resulting in an alcoholic beverage with favorable sensory characteristics. The pH of our beverage was 4.5, which is consistent with the fermentation process of alcoholic beverages, as it started from 5.74 (the pH of cubio). Due to the nature of the tuber, values as low as those in traditional wine were not achieved, but microbiological safety was ensured.
Sensory evaluation of the cubio beverage
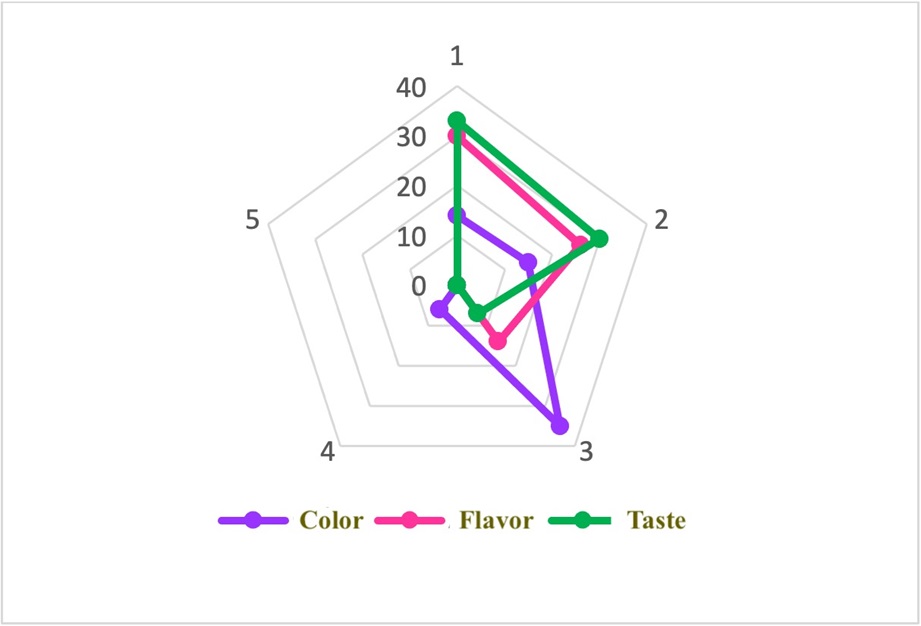
Fig. 2 shows the aspects rated in the sensory panel by the experts, with the highest score for flavor, followed by those for aroma and color. The panelists described the beverage as having a floral-fruity aroma and a semi-sweet flavor, indicating the presence of volatile compounds derived from fatty acids, carbohydrates, and amino acids produced in the biosynthetic pathway 45. This is because Saccharomyces bayanus has been linked to a higher yield of glycerol, lactic acid, succinic acid, malic acid, and, especially, acetate and ethyl ester, in comparison with other strains used in alcoholic fermentation such as Saccharomyces cerevisiae46. The floral aroma may be attributed to the fact that Saccharomyces bayanus can produce 2-phenylethanol (2-PE), a highly valuable aromatic alcohol with a rose-like aroma 30.
Regarding the color characteristics of the beverage, the panelists described it as bright lilac, which corresponds to values of 47.76±0.05 L*, 17.16±0.03 a*, and -23.84±0.08 b* in the CIELAB coordinate diagram. These values place the beverage in the red-blue chromatic zone with intermediate brightness, which is lighter compared to red grape wines. The positive L* value indicates a beverage with higher luminosity. The presence of acids in the beverage influences its color and aroma, since they act as antimicrobial agents during the production process 47. In comparison with the characteristics found in wines, pigmentation in the tuber beverage was achieved using 30-40 % of the pigments present in the raw material. Furthermore, copigmentation reactions involving hydroxycinnamic acids and flavonoids influenced the coloration of anthocyanins, contributing to the overall color profile 48. The aroma produced was linked to the alcohol content, acidity, dry extract, total and reducing sugars, and body of the wine 33.
The type of packaging is crucial to preserve the properties of wine, including its flavor, aroma, and overall quality, as appropriate materials can prevent oxidation and other chemical reactions that may degrade it over time. According to 49, selecting the right container is essential for maintaining the stability of bioactive compounds, particularly in wines derived from less conventional sources, as is the case of cubio.
As shown in Fig. 2, the values for each of the independent variables range between 1 and 3, demonstrating a good product acceptance. When the attributes were statistically correlated (data not presented), a low relationship was found between color and flavor and aroma, possibly due to a low correlation.
Figure 2: Sensory evaluation results obtained for the cubio alcoholic beverage. 1: I like it very much. 2: I like it slightly. 3: I neither like nor dislike it. 4: I dislike it slightly. 5: I dislike it very much.
Conclusions
Based on the experimental data, the cubio tuber was found to be a viable raw material for obtaining a fermented alcoholic beverage. The characterization of the Colombian blanca ojo morado variety of cubio revealed its potential as a valuable source of carbohydrates, including reducing sugars (2.18±0.002 %), starch (1.66±0.015 %), and proteins (4.48±0.064 %). These constituents served as substrates for yeast growth and subsequent fermentation. The stages of concentration and the incorporation of α-amylase and glucoamylase enzymes in the fermentation must increased the concentration of reducing sugars, providing a more favorable substrate for yeast metabolism and ultimately facilitating the production of ethanol.
The alcoholic beverage reached a pH value of 4.5, a total acidity of 7.21 ± 0.029 g.L−1, a density of 0.9947 mg.mL−1, and an alcohol concentration of 15.24 °GL. The concentration of reducing sugars experienced a gradual decrease, at a rate of 15.45 mg.L−1.day−1, and the production of alcohol reported a rate of 1.1786 °GL.day−1. All these values correspond to an optimal fermentation process. A sensory evaluation conducted by expert panelists yielded favorable ratings for the flavor and aroma characteristics of the beverage.
This suggests that the cubio tuber holds promise as an alternative ingredient for the production of high-quality alcoholic beverages, encouraging further exploration and research into the utilization of these ancestral tubers. Moreover, their high nutritional value underscores their potential as a promising food source for the future
References
License
Copyright (c) 2024 Martha Morantes Triana , Alfredo Lopez Molinello, Lena Prieto, Ismael Povea, Santiago Saenz

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
From the edition of the V23N3 of year 2018 forward, the Creative Commons License "Attribution-Non-Commercial - No Derivative Works " is changed to the following:
Attribution - Non-Commercial - Share the same: this license allows others to distribute, remix, retouch, and create from your work in a non-commercial way, as long as they give you credit and license their new creations under the same conditions.




2.jpg)