DOI:
https://doi.org/10.14483/23448393.23141Published:
2025-11-13Issue:
Vol. 30 No. 3 (2025): September-DecemberSection:
Environmental EngineeringUse of Computational Tools in the Modeling of Columns for Cr(VI) Adsorption in Wastewater by Means of Theobroma cacao L.
Uso de herramientas computacionales en el modelado de columnas para la adsorción de Cr(VI) en aguas residuales utilizando Theobroma cacao L.
Keywords:
Waste biomass, Adsorption column, Chromium (VI), Simulation, Water treatment, Wastewater treatment (en).Keywords:
Biomasa residual, Columna de adsorción, Cromo (VI), Simulación, Tratamiento de Aguas (es).Downloads
References
S. Bochynska et al., “The impact of water pollution on the health of older people,” Maturitas, vol. 185, art. 107981, Jul. 2024. https://doi.org/10.1016/J.MATURITAS.2024.107981
Rozirwan et al., “Ecological risk assessment of heavy metal contamination in water, sediment, and polychaeta (Neoleanira tetragona) from coastal areas affected by aquaculture, urban rivers, and ports in south Sumatra,” J. Ecol. Eng., vol. 25, no. 1, pp. 303–319, 2024. https://doi.org/10.12911/22998993/175365
A. R. Amal Raj, P. Mylsamy, V. Sivasankar, B. S. Kumar, K. Omine, and T. G. Sunitha, “Heavy metal pollution of river water and eco-friendly remediation using potent microalgal species,” Water Sci. Eng., vol. 17, no. 1, pp. 41–50, Mar. 2024. https://doi.org/10.1016/J.WSE.2023.04.001
S. Naik and S. E. Jujjavarapu, “Self-powered and reusable microbial fuel cell biosensor for toxicity detection in heavy metal polluted water,” J Environ. Chem. Eng., vol. 9, no. 4, art. 105318, Aug. 2021. https://doi.org/10.1016/J.JECE.2021.105318
G. Crini and E. Lichtfouse, “Advantages and disadvantages of techniques used for wastewater treatment,” Environ. Chem. Lett., vol. 17, no. 1, pp. 145–155, Mar. 2019. https://doi.org/10.1007/S10311-018-0785-9/METRICS
Z. Wang et al., “Overview assessment of risk evaluation and treatment technologies for heavy metal pollution of water and soil,” J. Clean. Prod., vol. 379, art. 134043, Dec. 2022. https://doi.org/10.1016/J.JCLEPRO.2022.134043
N. Zulfiqar, M. Shariatipour, and F. Inam, “Sequestration of chromium( vi ) and nickel( ii ) heavy metals from unhygienic water via sustainable and innovative magnetic nanotechnology,” Nanoscale Adv., vol. 6, no. 1, pp. 287–301, Dec. 2023. https://doi.org/10.1039/D3NA00923H.
A. Ayele and Y. G. Godeto, “Bioremediation of Chromium by Microorganisms and Its Mechanisms Related to Functional Groups,” J Chem, vol. 2021, no. 1, p. 7694157, Jan. 2021. https://doi.org/10.1155/2021/7694157
J. Jiang et al., “Utilizing adsorption of wood and its derivatives as an emerging strategy for the treatment of heavy metal-contaminated wastewater,” Environ. Poll., vol. 340, art. 122830, Jan. 2024. https://doi.org/10.1016/J.ENVPOL.2023.122830
Ministerio de Ambiente y Desarrollo Sostenible, “Resolucion 631 de 2015 vertimientos,” 2015. [Online]. Available: https://www.minambiente.gov.co/wp-content/uploads/2021/11/resolucion-631-de-2015.pdf
B. Gupta, A. Mishra, R. Singh, and I. S. Thakur, “Fabrication of calcite based biocomposites for catalytic removal of heavy metals from electroplating industrial effluent,” Environ. Technol. Innov., vol. 21, art. 101278, Feb. 2021. https://doi.org/10.1016/J.ETI.2020.101278
Y. Wang et al., “Research status, trends, and mechanisms of biochar adsorption for wastewater treatment: A scientometric review,” Environ. Sci. Eur., vol. 36, no. 1, pp. 1–17, Feb. 2024. https://doi.org/10.1186/S12302-024-00859-Z
F. Younas et al., “Current and emerging adsorbent technologies for wastewater treatment: Trends, limitations, and environmental implications,” Water, vol. 13, no. 2, art. 215, Jan. 2021. https://doi.org/10.3390/W13020215
S. Satyam and S. Patra, “Innovations and challenges in adsorption-based wastewater remediation: A comprehensive review,” Heliyon, vol. 10, no. 9, art. e29573, May 2024. https://doi.org/10.1016/J.HELIYON.2024.E29573
R. Yousef, H. Qiblawey, and M. H. El-Naas, “Adsorption as a process for produced water treatment: A review,” Processes, vol. 8, no. 12, art. 1657, Dec. 2020. https://doi.org/10.3390/PR8121657
M. Ince and O. Kaplan Ince, “An overview of adsorption technique for heavy metal removal from water/wastewater: A critical review,” Int. J. Pure Appl. Sci., vol. 3, no. 2, pp. 10–19, 2017. https://doi.org/doi.org/10.29132/ijpas.358199
A. F. Taiwo and N. J. Chinyere, “Sorption characteristics for multiple adsorption of heavy metal ions using activated carbon from Nigerian bamboo,” J. Mater. Sci. Chem. Eng., vol. 04, no. 04, pp. 39–48, 2016. https://doi.org/10.4236/MSCE.2016.44005
H. Sukmana, N. Bellahsen, F. Pantoja, and C. Hodur, “Adsorption and coagulation in wastewater treatment – Review,” Prog. Agri. Eng. Sci., vol. 17, no. 1, pp. 49–68, Nov. 2021. https://doi.org/10.1556/446.2021.00029
K. Erattemparambil, L. Mohan, N. Gnanasundaram, and R. Krishnamoorthy, “Insights into adsorption theory of phenol removal using a circulating fluidized bed system,” Arab. J. Chem., vol. 16, no. 6, art. 104750, 2023. https://doi.org/10.1016/j.arabjc.2023.104750
W. Liu et al., “Adsorption-based post-combustion carbon capture assisted by synergetic heating and cooling,” Renew. Sust. Energy Rev., vol. 191, art. 114141, 2024. https://doi.org/10.1016/j.rser.2023.114141
Y. Gao et al., “Interpretation of the adsorption process of toxic Cd2+ removal by modified sweet potato residue,” RSC Adv., vol. 14, no. 1, pp. 433–444, 2024. https://doi.org/10.1039/d3ra06855b
R. Lekshmi et al., “Adsorption of heavy metals from the aqueous solution using activated biomass from Ulva flexuosa,” Chemosphere, vol. 306, p. 135479, Nov. 2022. https://doi.org/10.1016/J.CHEMOSPHERE.2022.135479
R. M. Mohamed et al., “Adsorption of heavy metals on banana peel bioadsorbent,” J. Phys. Conf. Ser., vol. 1532, no. 1, art. 012014, Jun. 2020. https://doi.org/10.1088/1742-6596/1532/1/012014
T. Arsenie, I. G. Cara, M. C. Popescu, I. Motrescu, and L. Bulgariu, “Evaluation of the adsorptive performances of rapeseed waste in the removal of toxic metal ions in aqueous media,” Water, vol. 14, no. 24, art. 4108, Dec. 2022. https://doi.org/10.3390/W14244108
B. Siriweera and S. Jayathilake, “Modifications of coconut waste as an adsorbent for the removal of heavy metals and dyes from wastewater,” Int. J. Environ. Eng., vol. 10, no. 4, art. 329, 2020. https://doi.org/10.1504/IJEE.2020.110458
C. T. Tovar, Á. V. Ortiz, and M. J. Villadiego, “Remoción de cromo hexavalente sobre residuos de cacao pretratados químicamente,” Rev. UDCA Act. & Div. Cient., vol. 20, no. 1, pp. 139–147, 2017. https://doi.org/10.31910/rudca.v20.n1.2017.71
V. Marcantonio, E. Bocci, J. P. Ouweltjes, L. Del Zotto, and D. Monarca, “Evaluation of sorbents for high temperature removal of tars, hydrogen sulphide, hydrogen chloride and ammonia from biomass-derived syngas by using Aspen Plus,” Int. J. Hydrogen Energy, vol. 45, no. 11, pp. 6651–6662, 2020. https://doi.org/10.1016/j.ijhydene.2019.12.142
A. P. Sánchez, E. J. P. Sánchez, and R. M. S. Silva, “Simulation of the acrylic acid production process through catalytic oxidation of gaseous propylene using ChemCAD® simulator,” Ingeniare, vol. 27, no. 1, pp. 142–150, 2019. https://doi.org/10.4067/S0718-33052019000100142
U. Upadhyay, S. Gupta, A. Agarwal, I. Sreedhar, and K. Latitha, “Process optimization at an industrial scale in the adsorptive removal of Cd2 + ions using dolochar via response surface methodology,” Environ. Sci. Poll. Res., pp. 0–27, 2021. https://doi.org/https://doi.org/10.21203/rs.3.rs-811892/v1
A. Hameed, B. H. Hameed, F. A. Almomani, M. Usman, M. M. Ba-Abbad, and M. Khraisheh, “Dynamic simulation of lead(II) metal adsorption from water on activated carbons in a packed-bed column,” Biomass Convers. Biorefin., vol. 14, no. 7, pp. 8283–8292, Apr. 2024. https://doi.org/10.1007/S13399-022-03079-8/TABLES/3
D. Mohan and C. U. Pittman, “Activated carbons and low cost adsorbents for remediation of tri- and hexavalent chromium from water,” J. Hazard. Mater., vol. 137, no. 2, pp. 762–811, Sep. 2006. https://doi.org/10.1016/J.JHAZMAT.2006.06.060
B. Hemambika and V. R. Kannan, “Intrinsic characteristics of Cr6+-resistant bacteria isolated from an electroplating industry polluted soils for plant growth-promoting activities,” Appl. Biochem. Biotechnol., vol. 167, no. 6, pp. 1653–1667, Jul. 2012. https://doi.org/10.1007/S12010-012-9606-Y/METRICS
A. Agarwal, U. Upadhyay, I. Sreedhar, and K. L. Anitha, “Simulation studies of Cu(II) removal from aqueous solution using olive stone,” Clean. Mater., vol. 5, art. 100128, 2022. https://doi.org/10.1016/j.clema.2022.100128
J. Lara, C. Tejada, Á. Villabona, A. Arrieta, and C. Granados Conde, “Adsorción de plomo y cadmio en sistema continuo de lecho fijo sobre residuos de cacao,” Rev. ION, vol. 29, no. 2, pp. 113–124, Jan. 2016. https://doi.org/10.18273/REVION.V29N2-2016009
F. Benyahia and K. E. O’Neill, “Enhanced voidage correlations for packed beds of various particle shapes and sizes,” Part. Sci. Tech., vol. 23, no. 2, pp. 169–177, 2005. https://doi.org/10.1080/02726350590922242
A. G. Dixon, “Correlations for wall and particle shape effects on fixed bed bulk voidage,” Can. J. Chem. Eng., vol. 66, no. 5, pp. 705–708, 1988. https://doi.org/10.1002/cjce.5450660501
B. K. Koua, P. M. E. Koffi, and P. Gbaha, “Evolution of shrinkage, real density, porosity, heat and mass transfer coefficients during indirect solar drying of cocoa beans,” J. Saudi Soc. Agri. Sci., vol. 18, no. 1, pp. 72–82, 2019. https://doi.org/10.1016/j.jssas.2017.01.002
R. Ragadhita, A. Bayu, and D. Nandiyanto, “Curcumin adsorption on zinc imidazole framework-8 particles: Isotherm adsorption using Langmuir, Freundlich, Temkin, And Dubinin-Radushkevich models,” J. Eng. Sci. Tech., vol. 17, no. 2, pp. 1078–1089, 2022. https://jestec.taylors.edu.my/Vol%2017%20Issue%202%20April%20%202022/17_2_19.pdf
S. Nikam, D. Mandal, and P. Dabhade, “LDF based parametric optimization to model fluidized bed adsorption of trichloroethylene on activated carbon particles,” Particuology, vol. 65, pp. 72–92, 2022. https://doi.org/10.1016/j.partic.2021.05.012
M. R. Fouad, “Physical characteristics and Freundlich model of adsorption and desorption isotherm for fipronil in six types of Egyptian soil,” Curr. Chem. Lett., vol. 12, no. 1, pp. 207–216, 2023. https://doi.org/10.5267/J.CCL.2022.8.003
J. G. Amrutha, C. R. Girish, B. Prabhu, and K. Mayer, “Multi-component Adsorption Isotherms: Review and Modeling Studies,” Environ. Proc., vol. 10, no. 2, pp. 1–52, Jun. 2023. https://doi.org/10.1007/S40710-023-00631-0/FIGURES/2
T. Ahmad et al., “Enhanced adsorption of bisphenol-A from water through the application of isocyanurate based hyper crosslinked resin,” J. Mol. Liq., vol. 395, art. 123861, Feb. 2024. https://doi.org/10.1016/J.MOLLIQ.2023.123861
S. Nikam, D. Mandal, and P. Dabhade, “LDF based parametric optimization to model fluidized bed adsorption of trichloroethylene on activated carbon particles,” Particuology, vol. 65, pp. 72–92, Jun. 2022. https://doi.org/10.1016/J.PARTIC.2021.05.012
R. Fran Mansa, A. Osong Patrick, S. Kumaresan, and T. Ming Ling, “Simulation of lead removal using palm kernel shell activated carbon in a packed bed column,” 2021. [Online]. Available: https://easychair.org/publications/preprint/GZFQ
D. C. D. Caldeira, C. M. Silva, F. de Ávila Rodrigues, and A. J. V. Zanuncio, “Aspen Plus simulation for effluent reuse in thermomechanical pulp mills,” Water Sci. Tech., vol. 88, no. 3, pp. 751–762, Aug. 2023. https://doi.org/10.2166/WST.2023.242
A. Bringas, E. Bringas, R. Ibañez, and M. F. San-Román, “Fixed-bed columns mathematical modeling for selective nickel and copper recovery from industrial spent acids by chelating resins,” Sep. Purif. Tech., vol. 313, art. 123457, May 2023. https://doi.org/10.1016/J.SEPPUR.2023.123457
H. Patel, “Batch and continuous fixed bed adsorption of heavy metals removal using activated charcoal from neem (Azadirachta indica) leaf powder,” Sci. Rep., vol. 10, no. 1, pp. 1–12, 2020. https://doi.org/10.1038/s41598-020-72583-6
M. Hardyianto Vai Bahrun, Z. Kamin, S. M. Anisuzzaman, A. Bono, and M. H. V Bahrun, “Assessment of adsorbent for removing lead (pb) ion in an industrial-scaled packed bed column,” J. Eng. Sci. Tech., vol. 16, no. 2, pp. 1213–1231, 2021. https://jestec.taylors.edu.my/Vol%2016%20issue%202%20April%202021/16_2_23.pdf
M. L. R. M. Lubiano, C. V. L. Manacup, A. N. Soriano, and R. V. C. Rubi, “Continuous biosorption of Pb2+ with bamboo shoots (Bambusa spp.) using aspen adsorption process simulation software,” ASEAN J. Chem. Eng., vol. 23, no. 2, pp. 153–166, 2023. https://doi.org/10.22146/AJCHE.77314
How to Cite
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Download Citation
Recibido: 30 de diciembre de 2024; Aceptado: 10 de agosto de 2025
Abstract
Context:
Industrial growth and various anthropogenic activities have generated multiple pollutants, including heavy metals such as hexavalent chromium, or Cr(VI), which pose a major threat to both humans and the environment, as their characteristics make them persistent, bioaccumulative, and non-biodegradable.
Method:
In this paper, computer-aided process engineering (CAPE) was used to simulate an industrial-scale adsorption column packed with a biomass based on cocoa husk residues for the removal of Cr(VI) in solution. A parametric sensitivity analysis was conducted, using Aspen Adsorption as a simulation tool to analyze different column configurations.
Results:
The Freundlich isothermal model, in combination with the linear driving force (LDF) kinetic model, yielded efficient results in removing Cr(VI) via adsorption, with values of up to 97.1%. The best operating conditions included an initial concentration of 5000 mg/L, a bed height of 5 m, and an inlet flow rate of 100 m3/day.
Conclusions:
This study demonstrates that the use of computational assistance holds great potential for predicting the performance of an adsorption column packed with agro-industrial waste, which constitutes a safe and cost-effective alternative for the design and modeling of industrial-scale columns.
Keywords:
waste biomass, adsorption column, chromium (VI), simulation, water treatment, wastewater treatment.Resumen
Contexto:
El crecimiento industrial y diversas actividades antropogénicas han generado múltiples contaminantes, entre los que se encuentran metales pesados como el cromo hexavalente, o Cr(VI), los cuales representan una amenaza significativa tanto para el medio ambiente como para el ser humano, pues sus características los hacen persistentes, bioacumulativos y no biodegradables.
Método:
En este artículo se empleó la ingeniería de procesos asistida por ordenador (CAPE) para simular una columna de adsorción a escala industrial llena de biomasa a base de residuos de cáscara de cacao para la eliminación de Cr(VI) en solución. Se realizó un análisis de sensibilidad paramétrica, utilizando Aspen Adsorption como herramienta de simulación para analizar diferentes configuraciones de la columna.
Resultados:
El modelo isotérmico de Freundlich, junto con el modelo cinético de fuerza motriz lineal (LDF) arrojó resultados eficientes en la remoción de Cr(VI) mediante adsorción, con valores de hasta 97.1 %. Las mejores condiciones de operación incluyeron una concentración inicial de 5000 mg/l, una altura del lecho de 5 m y un caudal de entrada de 100 m3/día.
Conclusiones:
Este estudio demuestra que el uso de la asistencia computacional tiene gran potencial para predecir el rendimiento de una columna de adsorción llena de residuos agroindustriales, constituyéndose en una alternativa segura y rentable para el diseño y modelado de columnas a escala industrial.
Palabras clave:
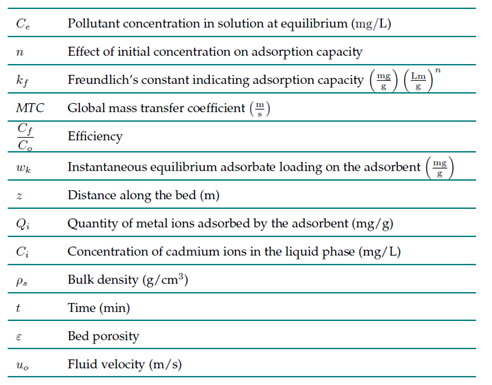
biomasa residual, columna de adsorción, cromo (VI), simulación, tratamiento de agua, tratamiento de aguas residuales.Nomenclature
Introduction
Water sources play an essential role in human life due to their great usefulness and versatility in different activities, e.g., industrial processes, agricultural and livestock activities, hospitals, shopping centers, rural areas, and domestic households, among others 1). Using this resource generates different types of wastewater that are loaded with pollutants. A large portion of this water, which originates from industrial activity, is considered to be of great concern due to its properties, as is the case with the toxic substances it contains 2)(3). This toxicity depends on various factors such as chemical composition, which varies from sector to sector, making the elimination of these substances a challenge, as each effluent contains unique pollutants that require customized treatment 4)(5). Among the various pollutants present in industrial wastewater are heavy metals, a worldwide concern due to their high toxicity, their detrimental impact on both the environment and human health, and their high strength and non-biodegradability 6).
Despite being an essential metal, chromium is considered to be one of the 16 main toxic pollutants with adverse effects on human health. This metal, which can be ingested, inhaled, or absorbed through the skin 7, is used in various manufacturing sectors, such as the metallurgy, tanning, cement, textile, and dyeing industries. These sectors represent major sources of pollution 8). Chromium ranks second in abundance among the heavy metal pollutants, surpassed only by lead. It is naturally found in three forms: metallic, trivalent, and hexavalent, with the latter, Cr(IV), being the toxic one. This form has a variety of detrimental health effects, such as digestive, urinary, reproductive, and immune system dysfunctions. Therefore, it is necessary to treat any water sources contaminated with Cr(IV) that are discharged into the environment 9). In Colombia, the limit for chromium in drinking water, as dictated by Law 0631 of 2015, is 0.5 mg/L 10, while the World Health Organization’s permitted level is 0.05 mg/L for Cr 11.
Adsorption is an effective method for removing unwanted pollutants, given its feasibility and easy scalability. This method is also able to effectively remove various inorganic chemicals that affect the adsorption system 12). This technique has several advantages that make it a very attractive method for use in water treatment. Among these advantages are its efficiency in removing various types of pollutants, including heavy metals, dyes, emerging contaminants, and other types of substances 13; its applicability at low temperature and pressure, since, compared to other water treatment processes, it does not require extreme conditions; its versatility in dealing with pollutants in both gaseous and liquid form 14; the regenerative capacity of the materials used in the adsorption process, which allows reusing biomaterials multiple times depending on the type of biomass employed and its characteristics 15; the speed at which this technique removes pollutants, allowing for effective and fast processes; and its versatility in enabling a modular and scalable design of the adsorption process in the form of columns or adsorbent beds, modeled on a small or a large scale as required 16.
This technique may involve one of two different adsorption methods: physisorption, wherein adsorption stems from the presence of weak attractions between the pollutant and the adsorbent material, generated by short-range electrostatic forces (i.e., van der Waals forces) 17; and chemisorption, where adsorption takes place due to the presence of chemical bonds between the adsorbate and the adsorbent, causing a stronger and more selective interaction. This process is less reversible than physisorption and requires considerably more energy for the desorption of the adsorbed species 18. The adsorption process can be carried out in two ways: in batches or in columns 19.
This technique has been recognized as one of the simplest and most economical strategies due to its remarkable ability for heavy metal removal 20)(21. Different biomaterials have been developed and used to this effect, such as Ulva flexuosa, a species of algae 22; babano shell 23; rapeseed (Brassica napus) 24; and coconut 25, among others. Cocoa (Theobroma cacao L.) is a widely cultivated product in Colombia, and it generates a large amount of residual biomass, comprising the husk of the cob, the husk of the bean, and the pulp. Among the different byproducts of this plant, the husk is a promising material for the production of adsorbent materials, given its abundance, low cost, and renewable nature. The cocoa husk is composed of cellulose, hemicellulose, and lignin, and it offers a high adsorption capacity, which makes it a sustainable alternative for the elimination of pollutants in aqueous media, e.g., heavy metals 26. Most adsorption studies have been conducted at the laboratory level under simple conditions and on small scales. This is due to different limiting factors, such as resource, space, and time availability, among others. In light of the above, several computational tools like Aspen Plus 27) or ChemCAD 28, have been developed for modeling processes on a larger scale, but scaling to solids in adsorption towers is still in its early stages. Therefore, researchers have searched for new ways to scale their proposals under the existing limitations. Among them, the Aspen Adsorption software has proven to be a tool for adsorption columns. This tool allows modeling multi-scale adsorption columns by simulating key phenomena such as mass transfer, adsorption equilibrium, and fluid dynamics. It allows predicting performance at the laboratory, pilot, and industrial scales, optimizing system design and operation based an experimental data. Its use in treating chromium-contaminated water also demonstrates its ability to respond to various operating conditions, improving process efficiency and supporting scaling from laboratory-level to industrial applications. This tool constitutes an advantage for a broader exploration of different parameters or operating variables such as flow, bed height, and pollutant concentration, among others, facilitating the optimization of the process before designing a pilot or building industrial plant.
A study published by 29) delved into the performance of dolochar as a Cd(II) adsorbent, using Aspen Adsorption to simulate a large-scale process. The results, obtained via the response surface methodology, show that, with optimal bed height, inlet concentration, flow rate, and fixed biosorbent mass values, the CD(II) ion adsorption capacity and depletion time of the packed dolochar bed are 1.85 mg/g and 11.39 hours, respectively.
Using Aspen Adsorption V11, the study by 30 simulated the adsorption of Pb(II) on tire-based activated carbon (TAC) and commercial activated carbon (CAC) in a fixed-bed column while considering different concentration ranges, bed heights, and flow rates. The optimal conditions found in this study included a concentration of 500 mg/L, a bed height of 0.6 m, and a flow rate of 9.88×10−4 m3/s, which yielded breakthrough times of 488 and 23 s for TAC and CAC, respectively, with removal capacities of 114.26 and 7.72 mg/g.
In light of the above, the objective of this study was to model an industrial column packed with Theobroma cacao L. for the adsorption of Cr(VI) in solution, using Aspen Adsorption to conduct a parametric sensitivity analysis and evaluate the performance of the system by altering key parameters. Our work demonstrates the potential of computational tools for predicting the performance of adsorption columns and provides a solid basis for the design and simulation of large-scale adsorption systems that employ agro-industrial materials for water treatment.
Materials and methods
Parameterization and modeling
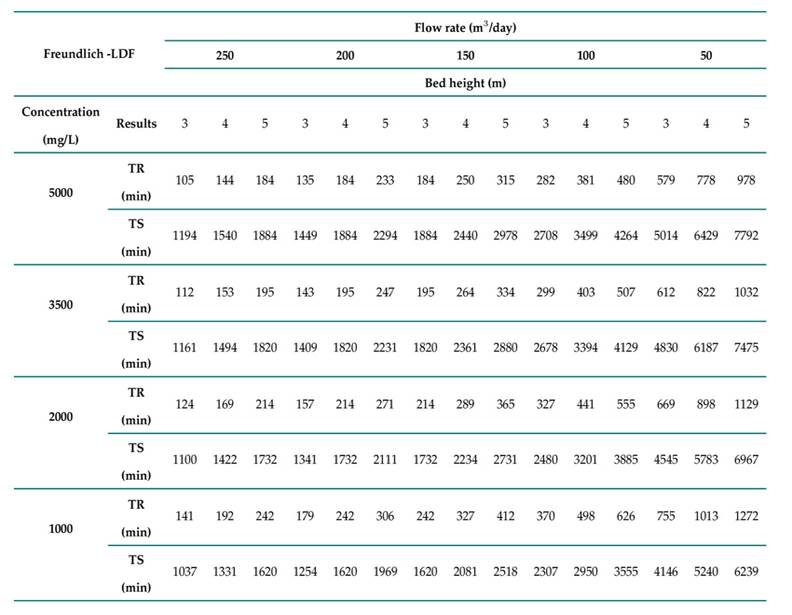
In this study, we used Aspen Adsorption V12.1 to simulate an adsorption column packed with Theobroma cacao L. for the removal of Cr(VI) in an industrial stream. A parametric study was carried out regarding the inlet flow rate, the initial concentration, and the column height, with the aim of determining the extent to which modifying these factors affects adsorption performance. We also conducted a sensitivity analysis that considered the breakthrough profile. Different parameter ranges were used to evaluate the performance of the adsorption system. For the inlet flow rate, values of 250, 200, 150, 100, and 50 m3/day were employed; for the bed height, we established values of 3, 4, and 5 mΣ30]; and, finally, for the initial concentration of Cr(VI), 5000, 3500, 2000, and 1000 mg/L were considered 31)(32.
To design our proposal, studies on the use of columns for the removal of heavy metals from industrial wastewater were used as a basis, considering the different parameters required to simulate the process in Aspen Adsorption. In this vein, we established a bed diameter of 1 m 33, a bulk density of 0.0365 g/cm3 for the biomaterial 34, a bed porosity of 0.67 (m3 of voids per m3 of bed), a total vacuum porosity of 0.4 35)(36, and a constant mass transfer coefficient of 1.37×10−4 s−1 (37.
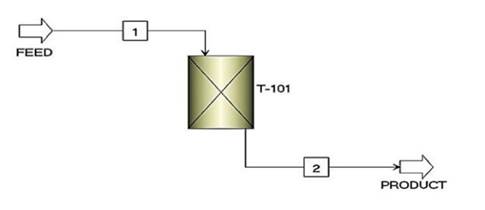
To understand the behavior caused by the interactions between the adsorbate and the adsorbent during the process, we employed the Freundlich isothermal model 38, while the linear driving force (LDF) kinetic model was used to determine the rate at which the adsorption occurred 39. Fig. 1 presents the simulation flowchart for the column.
Figure 1: Simulation flowchart for the adsorption column
Mathematical fundamentals
Mass balance
The equation used by Aspen Adsorption for the mass balance of the adsorption column is presented below:

Freundlich isothermal model
The Freundlich model considers a multilayer adsorption on a heterogeneous surface, where the distribution of the components depends on the time and energy of the secured sites 40)(41. This model is described by Eq. 2.

LDF kinetic model
The LDF kinetic model implies that the driving force for the mass transfer of components is a linear function of the concentration of the component in the liquid or solid phase 42)(43. This model is presented in Eq. 3.

Results and discusión
Evaluating the mathematical models using Aspen Adsorption
The Freundlich isothermal model and the LDF kinetic model were evaluated while considering the above-presented parameters and ranges. The results indicated that, by reducing the height of the column and increasing the flow rate, the break and saturation times can be reduced. This is due to the fact that, with a low bed height and a high flow rate, the fluid passes through the bed more quickly, which results in reduced process times. The results for the break time (TR) and the saturation time (TS) are shown in Table II.
Table II: Results obtained for the analyzed models
Influence of the inlet flow rate
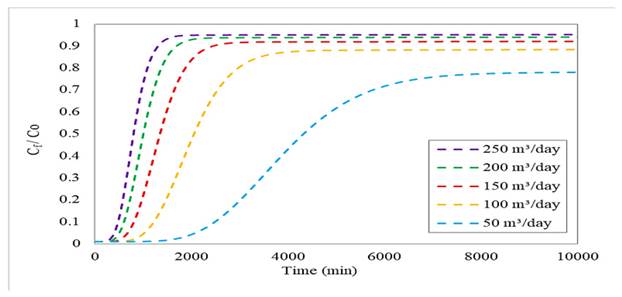
By means of a parametric sensitivity analysis, the influence of the inlet flow rate on the performance of the adsorption column was evaluated. To this effect, inlet flow rates of 250, 200, 150, 100, and 50 m3/day were considered. Fig. 2 shows the breakthrough profiles obtained from the Freundlich-LDF model. Note that the efficiency increases with the flow rate, but the breakthrough and saturation times are reduced. This behavior is due to the fact that a high inflow rate has a positive effect on mass transfer, causing a faster accumulation of the pollutant and decreasing the number of available active sites, which leads to reduced times. This behavior is also evident in the efficiency values obtained: 95.2 % for 250 m3/day, 94 % for 200 m3/day, 92.1 % for 150 m3/day, 88.4 % for 100 m3/day, and 78.1 % for 50 m3/day (44,45).
Figure 2: Breakthrough curve for a bed height of 5 m and an initial concentration of 500 mg/L at different flow rates
Influence of initial concentration
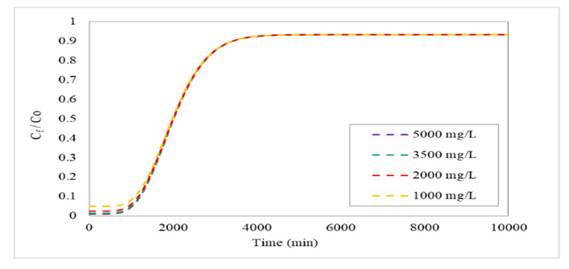
For the parametric sensitivity analysis of the initial Cr(VI) concentration, values of 5000, 3500, 2000, and 1000 mg/L were considered. Fig. 3 shows the breakthrough profiles obtained from the simulation of the Freundlich-LDF model. Note that, when this parameter is increased or decreased, the rupture times (TR) and saturation times (TS) obtained for each column configuration are very close to each other. Similarly, the efficiencies achieved show a similar trend, indicating that, under these conditions, the influence of concentration on the difference between TR, TS, and the overall system performance is limited. This phenomenon is due to the high presence of Cr(VI) in the flow, which causes an early break in the curve—this behavior is also observed at different times. Nevertheless, varying this parameter does not affect the adsorption performance, as the observed difference was less than 1 %. This can be attributed to different factors, such as the number of active sites available in the adsorbent, the strong affinity between the adsorbent and the adsorbate (which allows quickly reaching adsorption equilibrium), and the operating conditions considered, among others 46.
Figure 3: Breakthrough profiles for a column height of 5 m and an inlet flow rate of 100 m3/day at various Cr(IV) concentrations
Influence of bed height
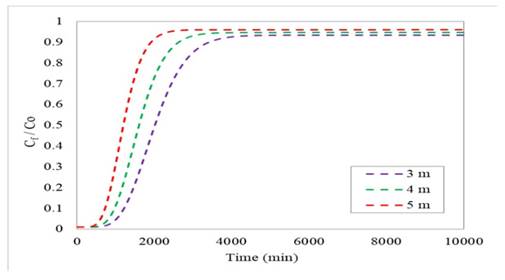
We also analyzed the influence of bed height on the performance of the adsorption column used for the removal of Cr(VI) in aqueous solution. The values considered in this analysis were 3, 4, and 5 m. Fig. 4 shows the breakthrough profiles obtained from the simulation of the Freundlich-LDF model. The breakthrough and saturation times increase with bed height, whereas efficiency is reduced. This is due to the fact that, with a larger adsorption surface, the fluid entering the column takes longer to exit. In addition, there are more active sites available, which extends the useful life of the adsorbent material, since it does not saturate as quickly, resulting in increased process times. On the other hand, the efficiency values obtained after the simulation were 92.9 % for 3 m, 90.6 % for 4 m, and 88.4 % for 5 m 47.
Figure 4: Breakthrough profiles for an initial concentration of 5000 mg/L and a flow rate of 100 m3/day at different bed heights
Comparison with other results in the literatura
The data obtained after conducting various simulations of the industrial cocoa-packed adsorption column were compared against the results of studies published in the specialized literature. It should be noted that this comparison has relative value, since each study was carried out under different inlet flow rate, initial concentration, bed height, and biomaterial conditions. The results of this work indicate that Theobroma cacao L. offers an acceptable in Cr(VI) removal. Table III shows de aforementioned comparison.
Table III: Comparison with literature-reported results

Conclusions
This study presented an innovative approach to modeling and simulating large-scale adsorption columns using agro-industrial waste (cocoa residues in this case) to remove heavy metals such as Cr(VI) from water systems. This work provides valuable quantitative data that contribute to the development and understanding of industrial adsorption processes. A parametric sensitivity analysis allowed evaluating the effect of varying the column’s bed height, inlet flow rate, and initial pollutant concentration on the efficiency of the process. The results that increasing the bed height increases the break and saturation times but reduces the adsorption efficiency. On the other hand, high inlet flows improve the adsorption efficiency but decrease the biomaterial’s saturation times. It is also noteworthy that the initial concentration does not have a significant effect on adsorption efficiency. These results constitute a robust technical basis for the design and optimization of industrial effluent treatment systems, as they allow anticipating the behavior of the system prior to its full-scale implementation.
Acknowledgements
Acknowledgements
The authors would like to thank Universidad de Cartagena for providing equipment, reagents, and technical and financial support for the project approved via Resolution no. 01385 and Minute no. 093 of 2021.
References
License
Copyright (c) 2025 Ángel D. González-Delgado, Ángel Villabona Ortiz , Candelaria Nahir Tejada Tovar

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
From the edition of the V23N3 of year 2018 forward, the Creative Commons License "Attribution-Non-Commercial - No Derivative Works " is changed to the following:
Attribution - Non-Commercial - Share the same: this license allows others to distribute, remix, retouch, and create from your work in a non-commercial way, as long as they give you credit and license their new creations under the same conditions.




2.jpg)